1. Introduction
Municipal solid waste management (MSWM) is a crucial service provided by cities all over the world, but it is often not managed well in developing countries. Improper solid waste management causes various forms of environmental pollution and health risks. Comparative studies of the various possible means of eliminating solid urban waste have revealed that the cheapest is landfilling [1]. On the other hand, generation of heavily polluted leachates having high organic and inorganic contents, presenting significant variations in both volumetric flow and its compositions in landfills are main environmental problems need to be controlled [2].
The composition of leachate greatly depends on the age of the landfill, waste type and hydrogeological factors [3]. According to the landfill age, leachate can be classified into three types: Young, middle-aged and stabilized [4]. Young landfill leachate due to its high organic content which are highly desirable for biodegradation. However, there is no unique method for leachate management to treat most of the compounds in a single step. A combination of physical (air stripping, membrane technologies), chemical (coagulation, flocculation and oxidation), biological methods (aerobic and anaerobic) and adsorption methods can be used to reach best solution [5–10].
Biological processes are low-cost method for landfill leachate, when compare with the others treatment options [11]. But, inhibitory compounds such as sulphate had negative effect on anaerobic degradation of landfill leachate. High concentrations of sulphur compounds in leachate hinder wastewater treatment as a biologically and it resulted that low production of methane gas. This phenomenon results from the anaerobic reduction of sulphates into sulphides. Excess amount of sulphate will promote enhanced growth of sulphate reducing bacteria [12]. Sulphides formed during the anaerobic process inhibit the growth of methanogens in the pH range of 7.2–8.5 [13, 14]. Sulphate content exceeds 1 g/L inhibition of methane production starts and total inhibition occurs when the sulphate exceeds 4.5 g/L [15]. High ratio of COD/SO4 such as 20:1 was found to be minimize inhibition effect on anaerobic methane production [16].
Ettringite is a hydrous calcium aluminum sulfate mineral with low solubility. Ettringite formation in high pH can be used to control sulphate reduction for anaerobic processes. Calcium, aluminium and sulphate react to form ettringite, according to Eq. (1):
Response surface methodology (RSM) is a mathematical and statistical technique that is useful for the optimization of chemical reactions such as ettringite crystallization. RSM is used in the development of an adequate functional relationship between a response of interest, and a number of associated control (or input) variables with limited number of experimental run. RSM designs offer ease of implementation and good performance over a wide range of applications [17–19]. RSM is a viable statistical method to optimize the treatment of industrial wastewaters [20, 21].
The purpose of this study is to investigate optimum conditions (important process variables such as Ca/SO4 and Al/SO4 molar ratios, and pH) for high sulphate removal efficiency (SO4−2 RE, %) with low operating cost (OC) ($/m3) by crystallization. The central composite design (CCD) was selected in this study because of its efficiency with respect to the number of runs required for fitting a second order response surface model [22]. And also, crystallinity, surface structure and other microscopic characteristics of sludge are characterized by scanning electron microscope (SEM) and energy dispersive X-Ray spectroscopy (SEM-EDX).
2. Materials and Methods
2.1. Chemical Composition of Landfill Leachates
Leachate samples were collected from the active detention pond which is less than 5 years old (classified as young landfill leachate) in municipal landfill site of Konya. The characteristics of studied landfill leachate are shown in Table 1.
Batch experiments were conducted in jar test apparatus for following minutes and rpms; 5 min at 120 rpm, 20 min at 45 rpm and finally 30 min for settling. Aluminum chloride (AlCI3) and calcium chloride (CaCl2) are used as the source of mineral formation and pH was adjusted with either 6M HCl or NaOH.
2.2. Experimental Design and Data Analysis
The CCD was created using Design Expert software (version 11.0.3, Statease Inc., USA). In this study, three important process variables, namely pH and molar ratios of Ca/SO4 and Al/SO4 were considered in the planning. CCD was applied for experimental studies augmented with six central replicates. In order to obtain optimum SO4−2 RE (%) was selected as the process response. The experimental ranges and levels of the independent variables used in RSM are given in Table 2, where a was set as 1.5 to determine second-order experimental points.
Independent variables and their levels for CCD and experimental results are given in Table 3. RSM postulates the functional relationship between the controllable input parameters and the obtained response surfaces [23, 24]. Response plots were generated using experimental data and the quality of the fitted polynomial model was expressed by the coefficient of determination R2 and its statistical significance was checked by Fisher’s F-test value and following by statistical significant model (p ≤ 0.05) by using analysis of variance (ANOVA).
ANOVA was used for three-dimensional plots and respective contour plots to obtain the interaction between the process variables (Ca/SO4 and Al/SO4 molar ratios and pH) and the responses (SO4−2 RE, % and OC, $/m3).
2.3. Analytical Methods
All chemicals used in this research were in analytical grade and supplied by Merck. COD, BOD5, Sulphate, Phosphate-phosphorus, Ammonium-nitrogen and pH were analyzed based upon Standard Methods [25]. Heavy metals were determined using the EPA 200.7 method of acid digestion followed by analysis by inductively coupled plasma with atomic emission spectroscopy (Optima 4300DV ICP, Perkin-Elmer, Boston, MA).
2.4. Scanning Electron Microscope (SEM)
SEM was performed using Evo LS10 (Carl Zeiss Microscopy GmbH) scanning microscope equipped with an energy dispersive X-Ray spectroscopy (EDX) Bruker EDX detector. The SEM images were recorded at 10 kV. The fractured sample was attached to a metal mount and sputter-coated with gold-platinum by using a sputtering machine (Cressington 108 auto, Cressington, UK).
3. Results and Discussion
3.1. Statistical Analysis
In recent years, statistical methods such as RSM, have been used to model, identify, analyze and establish the relationship of input and output process parameters. The traditional optimization approaches can only estimate the main process parameter effects and are not sufficient to express the interaction factor effects with minimum number of experiments and computational burden [26]. Therefore, process variables need to be varied simultaneously to estimate the contribution of interaction effects. In the present work, the relationship between the three appropriate process variables (pH, molar ratios of Ca/SO4 and Al/SO4) and the effects on the predicted responses (sulphate RE (%) and OC) was studied. The experimental data was analyzed by using the RSM procedure. CCD allows the development of second order quadratic regression models where each process response is assessed as a function of the process variables. The obtained results were then analyzed by ANOVA to examine the adequacy, correctness, and fitness of the obtained polynomial expressions. The second-order quadratic polynomial model for SO4−2 RE, % were subjected to the F-test to judge their relative significance.
The final mathematical model equation (Eq. (2)) was obtained by dropping insignificant model term using backward elimination procedure as following for in terms of actual factors:
The statistical parameters obtained from ANOVA for the established second-order quadratic polynomial regression model on the basis of the SO4−2 RE (%) are shown in Table 4.
In the present work, the proposed models to predict sulphate removal efficiency and treatment cost were significant at the 5% confidence level since p value was less than 0.05 and both R2 and adjusted R2 coefficients ensured a satisfactory adjustment of the quadratic model to the experimental data [27]. A high R2 coefficient is desirable and it expresses a good fit for the second order quadratic model to the experimental data [28, 29].
The variance coefficient (CV) is a measure of the model’s reproducibility and, when its value is less than 10%, the model can be considered as reasonably reproducible [30]. The adequate precision (AP) value is a measure of the ‘signal-to-noise ratio’ and was found as 18.7, indicating an adequate signal, because a value > 4 is desirable to use a regression model to navigate the space as defined by CCD [31, 32]. All other coefficients established by ANOVA postulate that the established regression model is significant and describe SO4−2 RE (%) satisfactorily.
Actual values were the result of an experimental run; however, the predicted values were determined from the model using the approximating functions. The data points of the actual response lie close to the predicted response as SO4−2 RE, % (Fig. 1).
3.2. Effect of Variables on the Sulphate Removal Efficiency
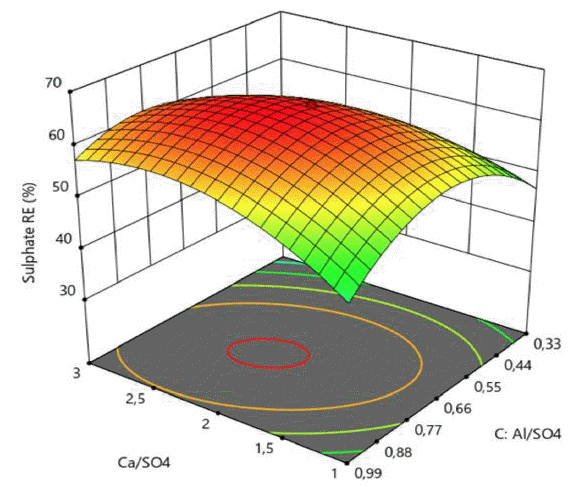
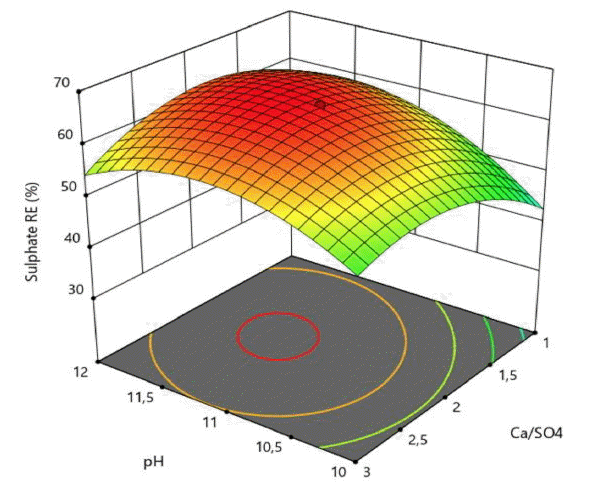
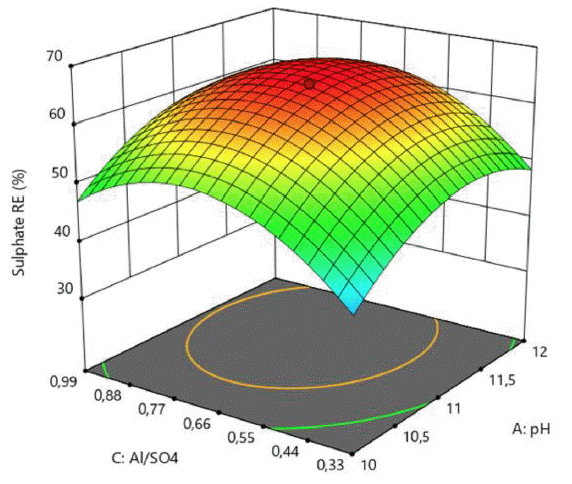
In order to gain a better understanding of the obtained results, the second order quadratic model was presented as 3D response surface contour plots in Fig. 2–4 on the basis of sulphate removal efficiency and treatment cost.
The RSM plots presented in Fig. 2–4 exhibit a curvature more or less noticeable depending on the significances of the interaction and quadratic model terms. Although some saddle points were observed, no true maximum points (for SO4−2 RE) and minimum points (for OC) could be detected on these plots, indicating that maximum SO4−2 RE (%) and minimum OC ($/m3) targets must be searched on the boundaries of the design space determined by experimental ranges of the process variables.
It is clearly seen that from the Fig. 2–4, the effects of Ca/SO4 and Al/SO4 molar ratios and pH were significant on sulphate removal efficiency. Sulphate removal efficiency sharply decreased at either highest or lowest molar ratios of Ca/SO4 and Al/SO4 values (Fig. 2).
The effects of pH on sulphate removal efficiency is more influential comparing to Ca/SO4 molar ratio changes (Fig. 3). Higher pH values and Al/SO4 ratios increased the sulphate removal efficiency (Fig. 4). Ettringite mineral is a stable above a pH of 10.7 and up to a pH of 12.5. Increasing the upper limit of pH at which ettringite is stable, ettringite is converted to calcium hydroxide, leaving sulphate and aluminate ions [33, 34].
3.3. Optimization Results
Numerical optimization was used to find the best solution by using design expert software. The maximum sulphate removal efficiency and minimum OC ($/m3) were considered to be two important goals in this study for the treatment of landfill leachate by the ettringite crystallization process. The corresponding importance of goals was selected as three out of five for all input variables and responses. The sulphate removal efficiency was estimated as 61.6% at pH 11.06, 1.87 of Ca/SO4 and 0.51 of Al/SO4 molar ratios with 0.52 $/m3 OC.
The desirability value was 0.77, showing that our model could be successfully applied to experimental data. The researchers have found different optimum conditions to form ettringite minerals in previous studies [35–37]. This is reveal that the composition of wastewater had an effect on stoichiometric ratio of each reagents and operating pH.
At the optimum condition, sulphate concentration decreased from 1,081 mg/L to 415 mg/L, but COD removal efficiency was only around 20%. Ettringite precipitation increased the ratio of COD/SO4 from 16:1 to 33:1 which was above the required ratio (COD/SO4 > 20) [16].
3.4. Application of SEM-EDX for Microstructural Analysis of the Solidified/Stabilized Chemical Sludge
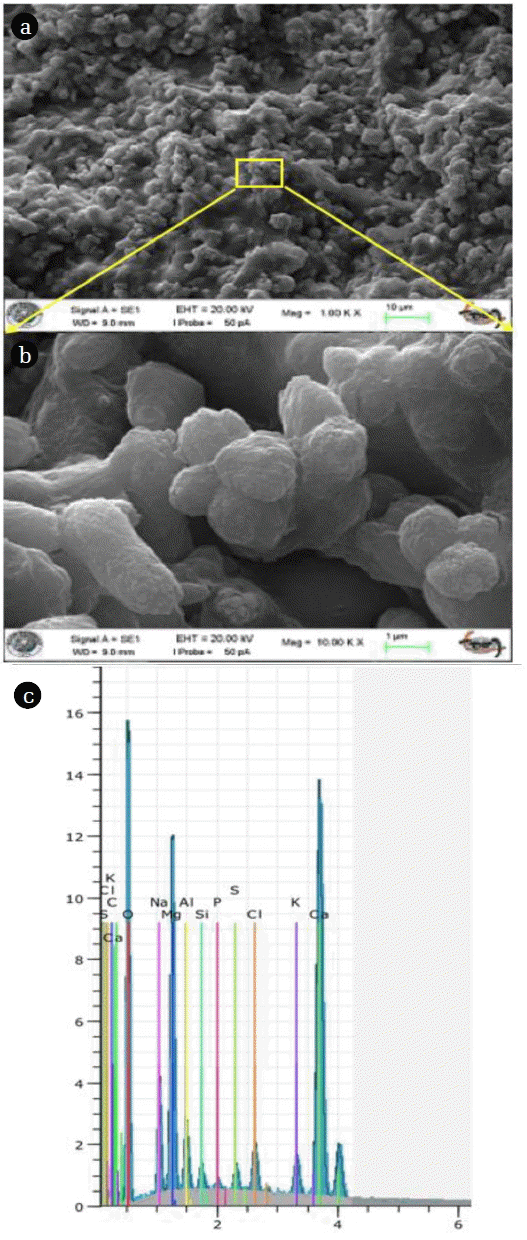
The microstructural studies of solidified/stabilized chemical sludge were conducted using SEM and SEM-EDX tests. As shown in Fig. 5, tricalcium aluminate and also, need-like microstructure of ettringite, a hydration by-product of tricalcium aluminate in the presence of sulfate ions were detected. Ettringite is a stable hydration product only while there is an ample supply of sulfate available [38].
EDX analysis shows that the principle elements present in a micron volume region are oxygen, calcium and sodium. Chemical composition of sludge is given in Table 5.
The presence of these elements and their distribution suggest that oxygen, the element most commonly found in the structure of the precipitate, is 65.98% in composition, calcium stems from the chemical substance added to precipitate, sodium comes from pH adjustment and due to the precipitated organic matter and carbonate minerals, the carbon atom is also observed at a relatively high rate. A minor amount of phosphorus, iron and silica ions is also visible for this study.
4. Conclusions
The optimum levels of Ca/SO4 and Al/SO4 molar ratios and pH variables were investigated by CCD and RSM in this study. Second order quadratic regression models were developed for the responses (SO4−2 RE, % and OC, $/m3). The present study clearly showed that CCD was a suitable method to optimize ettringite crystallization process by maximizing the SO4−2 RE (%) and minimizing the OC. The experimental data and model predictions were in good agreement. ANOVA resulted in high coefficients revealing that the proposed models were adequate and can be used to navigate the design space. At the optimum condition, COD/SO4 ratio increased from 16:1 to 33:1 resulted in less sulphate inhibition on anaerobic methane production. The ettringite crystallization process seems to be a very appropriate method for achieving the highest SO4−2 RE (%) and lowest OC for landfill leachate.