AbstractCheonsu Bay, one of the most important in Korea as a coastal fishery is a semi-enclosed bay that is surrounded by large farmlands and industrial areas. This coastal environment has been affected by anthropogenic pollutants, such as polychlorinated biphenyls (PCBs) and organochlorine pesticides (OCPs). The objectives of this study were to investigate the distribution of PCBs and OCPs in sediment, Manila clams, and mussels from Cheonsu Bay; the accumulation pattern of these chemicals in these bivalves in relation to seasonal changes; and the ecological risk from sediments and the risk to the Korean population from the consumption of these bivalves. The levels of ∑PCBs, ∑DDT, and ∑HCHs were 69.3–109, 40.3–49.3 and 6.25–17.8 ng/g lipid in Manila clams, and 70.6–159, 38.6–102 and 9.00–13.5 ng/g lipid in Mussels. Significant seasonal variations in PCBs and OCPs concentrations were observed in the two bivalves, suggesting that the accumulation of PCBs and OCPs in these species is related to their spawning times. The dietary intake of these two bivalves and the resulting lifetime cancer risk (LCR) and non-cancer risk were calculated for the human population. The consumption of these bivalves seemed to be safe in relation to human health with negligible LCR and non-cancer risk.
1. IntroductionPolychlorinated biphenyls (PCBs) and organochlorine pesticides (OCPs), which are anthropogenic organic pollutants, have been considered important contaminants for several decades. PCBs and OCPs are hydrophobic and lipophilic substances that persist in the environment for very long periods [1]. Because of the extreme stability of many of their isomers, PCBs were widely used in many products until they were banned in developed countries during the late 1970s. OCPs were major agricultural pesticides until they were restricted because of their serious side effects. Chronic toxicity, reproductive, carcinogenicity, and develomental toxicity, as well the biochemical effects of comecial PCBs mixture and OCPs have been investigated in fish and wildlife species [2–6]. As well, ecological risk, including bio-magnification, bio-accumulation of those pollutants in food web in marine environment was also reported [7–8].
As a result of much effort, including restrictions, the release of PCBs and OCPs to the environment has decreased over the last few decades. However, because of the hydrophobic nature of these compounds, huge amounts have sedimented to the sea floor and have been adsorbed onto organic particles [9]. Contaminated marine sediment and suspended organic particles can be a secondary source of organic pollutants to marine organisms, particularly sediment-dwelling organisms [10–12]. Because of their high octanol/water partition coefficient (Kow), these hydrophobic organic pollutants accumulate in the bodies of marine organisms [9]. Even extremely low concentrations of bioaccumulative pollutants detected in water or bottom sediments may result in their accumulation in marine organisms such as fish and shellfish [13]. Bivalves have been widely used to obtain information about local pollution [14, 15], and were recommended as bio-indicators by international conventions such as the OSPAR Commission, the Barcelona Convention, and the Helsinki Commission [16–18].
Manila clams (Ruditapes philippinarum) and mussels (Mytilus coruscus) are widely distributed throughout the intertidal zones along the western coast of Korea [19]. Mussels live on rocky shores, attaching themselves to rocks or to each other by means of byssal threads. As part of the Mussel Watch Program in Korea, Kim et al. assessed the contamination levels and accumulation features of PCBs and OCPs along the entire coast of Korea [20]. Ramu et al. analyzed mussels from 20 coastal locations in Korean waters to determine the concentrations of PCBs and OCPs [21]. Manila clams are also regarded as a useful bioindicator of sediment pollution because they live in sandy-mud bottoms and extrude their siphons to filter feed from near-bottom water [22]. The bio-accumulation of organic pollutants in Manila clams has been reported in recent studies [14, 23, 24].
Bioaccumulated hydrophobic organic pollutants are associated with the lipid content of biota. Lipids and fats change throughout the seasons in relation to an animal’s physiological condition, such as spawning and gametogenesis [25, 26]. Thus, the accumulated concentrations in biota of lipophilic contaminants, including organochlorine contents, can be influenced by seasonal changes and age-grade. Both Manila clams and mussels are commercially important bivalves, because they are a fishery food resource in Korea. Thus, understanding the bioaccumulation mechanisms in these aquatic organisms is critical to predicting their potential toxicity to human health [27]. However, the effects of season and size on organochlorine concentrations in R. philippinarum and M. coruscus collected in Korea have not been previously reported.
Cheonsu Bay, which is one of the most important bays in Korea, has a long tidal beach with a length of about 2,000 km. Both sandy-mud flat and tidal rock areas, the habitats of Manila clams and mussels, respectively, are well developed along the shoreline of Cheonsu Bay. This area is surrounded by agricultural and industrial areas [28], and the environmental pollution and ecological changes by anthropogenic activities have been reported [29]. Park et al. reported the environmental impacts on water quality of Cheonsu Bay derived from the input of adjacent fresh waters [30].
In order to understand the accumulation characteristics of PCBs and OCPs in Manila clams and mussels, surface sediment, Manila clams, and mussels from Cheonsu Bay, Korea, were collected. The objectives of this study were thus (1) to investigate the current distributions of PCBs and OCPs in sediment, Manila clams, and mussels; (2) to determine how the accumulation patterns of these chemicals in Manila clams and mussels are related to seasonal changes; and (3) to assess the ecological risk from the sediment and the risk to the Korean population posed by consuming these two bivalves.
2. Materials and Methods2.1. Sample Collection
Fig. 1 shows three sampling sites from Cheonsu Bay, Korea. These sampling sites were selected in order to provide full coverage of this bay. Surface sediments, Manila clams, and mussels were collected from these three sampling sites in different seasons.
Intertidal sediment samples were collected with a stainless steel spoon. Sediment samples were collected in glass bottles and stored frozen until they were freeze-dried using Labconco Freezone 6.
Manila clams were hand collected in December, May, and August in 2001–2002, and mussel samples were collected in December, May, and July in 2001–2003, in order to compare the accumulation patterns of PCBs and OCPs in the tissues of these two bivalves according to the sampling season and size. The collected bivalves were wrapped in aluminum foil and stored frozen until analysis. The shells of the bivalves were removed, and the soft tissues, including the viscera, were pooled and homogenized for analysis. All of the samples were freeze-dried using Labconco Freezone 6. The samples were analyzed for PCBs and OCPs.
2.2. Chemical AnalysisThe extraction and analysis of PCBs and OCPs were conducted according to the method described by Lauenstein and Cantillo [31], with some modifications. Freeze-dried sediment and pooled clam tissue samples were spiked with 2,2′,4,5′,6-pentachlorobiphenyl (PCB 103), 2,2′,3,3′,4,5,5′,6-octachlorobiphenyl (PCB 198), and 4,4′-dibromo octafluoro biphenyl (DBOFB) as surrogate standards, and a Soxhlet extraction was carried out using hexane:acetone (1:1, v/v). Sulfur was removed from the extracts of the sediments using activated copper (Granular, 20–30 mesh, J.T. Baker Co., USA). Each extract was passed through a column containing deactivated silicagel (~60 mesh; Merck, Germany)/alumina (~150 mesh; Aldrich Chemical Co., USA) withhexane:acetone (1:1, v/v) for cleanup to remove the polar/non-polar interference from the extracts. The extracts was also cleaned using a deactivated Florisil® (60–100 mesh; J.T. Baker Co., USA) column with hexane for further separation of PCBs from the most extracted components. Samples were concentrated to 0.5 mL under nitrogen and sealed in vials until analyzed.
2.3. Instrumental Analysis and Quality ControlA gas chromatograph equipped with a 63Ni electron capture detector (GC-ECD Hewlett Packard 5890) was used to identify and quantify the PCBs and OCPs. The GC column used for the analysis was a DB-5 (25-μm bonded phase; J&W Scientific Co Ltd., USA). The organic carbon in the sediment sample was quantified using a Carlo Erba CNS elemental analyzer 1106 following the method described by Hedges and Stern [32]. The lipid weight percentages (weight/weight basis) in the tissues of the organisms were operationally determined using the method of Qian et al. [33]. The same process and analytical method were used for blanks without samples to determine the contamination during the analytical process, and all the presented results were corrected for the blank levels. All of the glassware was cleaned with detergent, baked at 450°C for 4 h, and rinsed with hexane before use. The recovery percentages of the spiked PCBs and OCPs varied from 75.4% to 116.2% for the sediments, and 65.6% to 123.3% for the biological samples. The method detection limits (MDL) of the procedure for each compound ranged between 0.01 and 0.03 ng·g−1dw. Concentrations detected at less than the MDLs were expressed as not detected (ND). A reagent blank was used to determine laboratory contamination. To check for interference, a spiked sample consisting of all the chemicals was run. For duplicate samples, the average reproducibility of the method varied by less than 10%. Quality assurance procedures were performed by co-analyzing standard reference materials (SRM) of the appropriate matrix, namely, the sediment NIST 1941b and mussel tissue homogenate IAEA-142, with recoveries ranging from 62% to 123%. The 18 congeners (PCBs 8, 18, 28, 29, 44, 52, 66, 87, 101, 105, 110, 118, 128, 138, 153, 170, 180, and 187), including 13 major components in environmental mixtures [34], were analyzed. ∑PCB estimates were derived from the sum of the measured 18 congeners multiplied by a standard factor of two used by NOAA NS&T in 1987 and Howell et al. (2008). The ∑DDT and ∑HCH concentrations were found using the sums of six metabolites (o,p′-DDE, p,p′-DDE, o,p′-DDD, p,p′-DDD, o,p′-DDT, and p,p′-DDT) and four isomers (α-HCH, β-HCH, γ-HCH, and δ-HCH), respectively.
2.4. Calculation of Human Health Risk from Dietary IntakeTo assess the human health risk from ingesting Manila clams with PCBs and OCPs, the exposure levels (expressed as average daily intakes (ADI)) for PCBs and OCPs were calculated. The average daily consumptions of Manila clams (0.80 g/d) and mussels (0.99 g/d) for the general population in South Korea were obtained from the Food Balance Sheet Korea published by the Korean Rural Economic Institute [35]. Lifetime cancer risk (LCR) and hazard indices (HI) were derived using the calculated ADI in this study, the cancer slope factor (CSFo), and the oral reference dose (RfDo) for each compound, following the method of health risk assessment suggested by the National Research Council (NRC) and National Academy of Science (NAS) of the USA [36].
Here, Cc is the concentration of organochlorine pesticide (ng·g−1ww) of the clams, IRc is the ingestion rate per unit of time (kg·d−1), BW is the body weight of a human (65 kg), ED is the exposure duration (70 y), EF is the exposure frequency (365 y−1), and AT is the average time for exposure (365 d·y−1 for 70 y). The cancer slope factor and oral reference dose were obtained using an integrated risk information system (IRIS) reported by the US EPA [37].
2.5. Statistical AnalysisAll of the statistical analyses were performed using the PASW 18. One-way ANOVA, Hierarchical cluster analysis (HCA) and principle component analysis (PCA) were used to explain the spatial distributions of the PCBs and OCPs in the sediment samples. Pearson’s correlation test was applied to consider the correlation between the contaminants in the sediments and contaminants in the clams, and between Log Kow and Log BSAF for organic pollutants. The levels of significance were 0.05.
3. Results and Discussion3.1. PCBs and OCPs in Sediment from Cheonsu BayOrganochlorine pollutants such as PCBs, DDTs, and HCHs were detected in all of the sediment samples collected in Cheonsu Bay. During the sampling period, the mean concentrations of ∑PCBs, ∑DDTs, and ∑HCHs in the sediments collected from sites 1, 2, and 3 were 0.86 ± 0.12, 0.22 ± 0.035, and 0.20 ± 0.051 ng/g dry weight, respectively (Table 1). The organic carbon contents in the sediments were 0.32%, 0.23%, and 0.42%, respectively. In order to verify spatial and temporal homogeneity of chemical sediment parameters, including the concentration of contaminants during the sampling period, one-way ANOVA was performed for sediment groups collected from all sampling stations. There was no statistical difference in the organochlorine pollutant levels in the sediments collected at the three sampling stations, indicating no seasonal or special difference in the organochlorine pollutant levels in the sediments during the period (F = 0.41, p = 0.67) (Supplementary data 1). Therefore, collected sediments from these sampling stations were suitable for representing the pollution level of Cheonsu Bay with PCBs and OCPs.
Anthropogenic pollutants, including industrial and agricultural chemicals from the watershed, have been potential sources of PCBs and OCPs in Cheonsu Bay. According to the Korea Agricultural and Rural Infrastructure Corporation [39], the numbers of industrial factories and oil consumption in the area surrounding Cheonsu Bay were much higher than those of other areas in Korea. The total amount of DDT applied onto the areas adjacent to Cheonsu Bay from 1946 to 1974, was approximated to be 36.72 tons [40, 41].
In Korea, the use of PCBs in dielectric fluid was restricted in 1979, and the import of PCBs was prohibited in 1984 [42, 49]. Despite this ban, high levels of PCBs have been found in some coastal areas near industrial complexes or large harbors, as listed in Table 2a. However, according to the total PCB estimates (Table 1), none of the sites in this study exceeded the minimum values reported for the sediment quality guidelines, including the effects-range-low (ERL) for marine sediments (22.7 ng/g dry weight) [43]. As listed in Table 2a, the PCB and OCP levels in the sediments of this study area were comparably lower than those reported in other polluted coastal areas [50, 51].
Fig. 2 presents the profiles of the PCBs and OCPs in the sediments from Cheonsu Bay. The most important homologue group of PCBs found in the sediments appeared to be tetra-CBs, which accounted for about 30% of the PCB contamination, followed by penta-CBs, which accounted for 27%. In order to consider the source of PCB pollution, the congener profiles of PCBs in this area were compared with those of commercial PCB mixtures. Typical chlorination level plots from the sediment samples and PCB commercial mixtures such as those of Aroclor 1248, 1254, and 1260 are illustrated in Fig. 2a. All the profiles indicated that the PCBs in the sediments were of a composite mixture and that they derived from more than one technical formulation with different degrees of chlorination (Fig. 2b). Similar PCB profiles were also estimated in other areas of Korea [24], and their results showed that the PCBs in Nakdong River sediment were influenced by Aroclor 1254 and 1260.
As shown in Fig. 2c and 2d, p,p′-DDD and β-HCH were the dominant pesticides among the OCP compounds in the sediment. As listed in Table 1, the DDT compositional ratio (p,p′-DDT/p,p′-DDE+p,p′-DDD) of the sediment samples had a range of 0.25–0.71 in this area. This result suggests that the DDTs found in the studied sediment were mainly weathered DDTs from past use. The α-HCH/γ-HCH ratios in the sediment samples were below one, indicating the previous use of lindane (γ-HCH) in this area [56]. The Stockholm conventions classified HCHs, including α-HCH, β-HCH, and lindane (γ-HCH), as “Additional POPs” in 2009, because of their bioaccumulation, their potential for long-range transport, and their adverse effects. The technical HCH mixture has been banned in many countries since the 1970s and 1980s, while lindane has been used until recently.
Distributions and seasonal changes in PCBs and OCPs in Manila clams and mussels collected in Cheonsu Bay.
The lipid-normalized concentrations of PCBs and OCPs in the Manila clams and mussels from Cheonsu Bay, Korea, are summarized in Table 3. The PCB levels were the highest in the tissues of the Manila clams and mussels, with concentrations of 91.2 ± 20.2 ng/g and 118 ± 36.4 ng/g lipid weight, respectively. The concentrations of DDTs in the Manila clams and mussels were 45.5 ± 4.68 ng/g and 68.8 ± 31.8 ng/g lipid weights, respectively. The HCH concentrations in the Manila clams and mussels were 12.9 ± 5.95 ng/g and 11.2 ± 2.25 ng/g lipid weights, respectively, showing lower levels than the DDTs. These levels were relatively lower than those previously reported for Manila clams and mussels collected in other coastal areas in the world, showing that seafood in Korea is less contaminated than that in other coastal areas (Table 2b). Although the consumption of HCHs was higher than that of DDTs in northeast Asian countries [62], the accumulation levels of HCHs were lower than those of the DDTs in the Manila clams and mussels collected from the study area. This may be the result of the higher degradability and lower lipophilicity of HCHs than those of DDTs [63].
The values of PCBs and OCPs in the Manila clams that were 25–45 mm in size and in the mussels that were 30–70 mm in size, collected at different sampling times are compared in Fig. 3. The mean of the lipid normalized ∑PCB concentrations in the Manila clams collected in May and in mussels collected in July were significantly lower than those collected in December (one-way ANOVA, p < 0.05). The lowest lipid normalized ∑HCH and ∑DDT concentrations in the Manila clams and mussels were also observed in May and July, respectively. In addition, the mean of the lipid normalized ∑HCH concentration in the Manila clams collected in May was significantly lower than those for clams collected in other periods (one-way ANOVA, p < 0.05).
This result might be related to a seasonal physiological change in these organisms, and its possible influencing factor was reproduction. The ∑PCB, ∑DDT, and ∑HCH values in the Manila clam tissues in May decreased by 36.6%, 14.3%, and 56%, respectively, compared with those for December. In the case of the mussels, the ∑PCB, ∑DDT, and ∑HCH values in the tissues collected in July decreased by 38.4%, 58.3%, and 23.7%, respectively, compared to those obtained in May. In this area, the spawning period of Manila clams is from April to June [64], and that of mussels is from May to July. This suggests the possibility of release of organochlorines (OCs) from the bodies of Manila clams and mussels through spawning behavior.
Other studies found similar seasonal variation in OC accumulation in the bodies of aquatic organisms, including fish [65, 66], cetacean [67], rainbow trout [68], and the copepod Acartia tonsa [69]. Moreover, Hummel et al. reported that mussels might “lose” one-half to two-thirds of their PCB content by means of reproductive output [70]. Because the concentrations of OCs in Manila clams and mussels is able to be varied by their breeding behaviors, seasonal consideration in using these bivalves as an indicator species for risk assessment is more required. So we investigated that how risks to human health change with consuming these two bivalves as a result of seasonal variation of OCs in those bivalves.
Comparison of organic pollutant accumulation potentials of Manila clams (Ruditapes philippinarum) and mussels (Mytilus coruscus) and analysis of human health risk posed by consuming these two bivalves
The organic pollutant accumulation potentials of Manila clams, Ruditapes philippinarum, and mussels, Mytilus coruscus, were compared, and their PCB and OCP concentration profiles are shown in Supplementary data 2. The tetra-, penta-, and hexa-CBs were the major homolog groups in the Manila clams and mussels, constituting 67% and 77% of the total PCBs in Manila clams and mussels, respectively. The predominant congeners of PCBs were PCB 138 and 153 in both Manila clam and mussel tissues. In previous studies of other organisms in Korea, similar profiles were reported for porpoise, fish, and human serum [67, 71, 72]. The other major congeners were PCB 52, 87, and 44 in Manila clams and PCB 110, 52, and 44 in mussels. Among the OCPs, the major compound was p,p′-isomers of DDT followed by β-HCH, indicating long-term degradation from the use of those OCPs.
The accumulation patterns of the OCs in Manila clams were slightly different from those in mussels. In order to compare the accumulations of OCs in these two bivalves, a hierarchical cluster analysis (HCA) of the important pollutants in Manila clams and mussels collected at different sampling times, and of the sediments, was carried out (Fig. 4). As a result of this HCA, Manila clams and sediment were included in Group 1, and the other cluster, Group 2, consisted of mussel samples. This result indicated that the distributions of PCBs and OCPs in Manila clams were more closely related to those in sediment than those in mussels. This result might be caused by the habitat difference between these two species. Mussels inhabit the reefs of coastal regions, attached to rocks. On the other hand, Manila clams dwell on the sediment surface. Because Manila clams bury their body in a sandy-mud bottom and filter-feed using a siphon [22], they have a greater chance of being exposed to the bottom sediment than do mussels.
To estimate the human health risk from the ingestion of Manila clams and mussels collected from this area, the dietary intake and resulting LCR and non-cancer risk were calculated. The LCRs from ∑PCBs, ∑DDTs, and HCHs posed by the consumption of Manila clams and mussels during the sampling period are reported in Table 4. During the sampling period, the highest LCR and HI for these OCs in Manila clams were observed in August. On the other hand, the highest LCR and HI for these OCs in mussels were observed in December. The LCR and HI of these OCs, except for β-HCH, induced by the annual consumption of Manila clams were lower than those of mussels. Although there was a sharp fluctuation in the human health risk value, the risk levels did not exceed the acceptable risk (1 in 100,000), indicating that the consumption of these two bivalves from Cheonsu Bay seems to be safe in relation to human health, with negligible LCR and non-cancer risk.
4. ConclusionsWe investigated the concentrations of PCBs and OCPs in sediments and bivalves (Manila clams and mussels) collected from Cheonsu Bay, Korea. PCBs and OCPs were detected in most samples, but their levels were relatively lower than those in the contaminated marine areas. Seasonal variations of these OC levels in the two species were observed. The lowest levels of these OCs were found in Manila clams in May and in mussels in June. The accumulation patterns of these chemicals in both bivalves were related to their habitats and behavior, such as spawning. The results of this study showed that the consumption of Manila clam and mussels collected in Cheonsu Bay seems to be safe in relation to human health according to the calculated LCR and HI.
Although this bio-monitoring study gave information on the behavior and fate of PCB and OCPs in the environment of Cheonsu Bay, the long-term monitoring would need to be continued in order to understand the recent temporal changes in this estuarine environment and the biological fate of these chemicals in Manila clams and mussels.
SUPPLEMENTARY MATERIALOne-way ANOVA Results for HCHs, DDTs, and PCBs Concentrations in Sediment from Cheonsu Bay (α = 0.05)
SUPPLEMENTARY MATERIALProfiles of a) PCBs and b) OCPs concentrations in Manila clams and mussels from Cheonsu Bay.
AcknowledgmentsThis study was supported by a grant from the Korean Ministry of Land, Transport and Maritime Affairs (Marine Biotechnology Program) allocated to Kyung-Hoon Shin and partially supported by a grant from the Ministry of Ocean and Fisheries of Korea (through the project Development of sustainable remediation technology for marine contaminated sediments, PM 58230) of the Korea Institute of Ocean Science and Technology.
References1. Tolosa I, Bayona IM, Albaiges J. Aliphatic and polycyclic aromatic hydrocarbons and sulfur/oxygen derivatives in Northwestern Mediterranean sediments: Spatial and temporal variability, fluxes and budgets. Environ Sci Technol. 1996;30:2495–2503.
2. Garcia-Reyero N, Denslow ND. Applications of genomic technologies to the study of organochlorine esticide-induced reproductive toxicity in fish. J Pesticide Sci. 2006;31:252–262.
3. Lee SH, Ra JS, Choi JW, Yim BJ, Jung MS, Kim SD. Human health risks associated with dietary exposure to persistent organic pollutants (POPs) in river water in Korea. Sci Total Environ. 2014;470/471:1362–1369.
4. Hansen DJ, Parrish PR, Lowe JI, Wilson AJ, Wilson PD. Chronic toxicity, uptake, and retention of aroclor 1254 in two estuarine fishes. Bullet Environ Contam Toxicol. 1971;6:113–119.
5. Hatanaka J, Doke N, Harada T, Aikawa T, Enomoto M. Usefulness and rapidity of screening for the toxicity and carcinogenicity of chemicals in medaka, Oryzias latipes. Jpn J Exp Med. 1982;52:243–253.
6. Ulbrich B, Stahlmann R. Developmental toxicity of polychlorinated biphenyls (PCBs): A systematic review of experimental data May 2004. Arch Toxicol. 2004;78:252–268.
7. Hu G, Dai J, Mai B, et al. Concentrations and accumulation features of organochlorine pesticides in the Baiyangdian Lake freshwater food web of North China. Arch Environ Contam Toxicol. 2010;58:700–710.
8. Gómez-Gutiérrez A, Bayona JM, Albaigés J. Screening ecological risk assessment of persistent organic pollutants in Mediterranean Sea sediments. Environ Int. 2007;33:867–876.
9. Magnusson K, Ekelund R, Grabic R, Bergquist PA. Bioaccumulation of PCB congeners in marine benthic infauna. Mar Environ Res. 2006;61:379–395.
10. Tanabe S, Tatsukawa P, Phillips DJH. Mussels as bioindicators of PCB pollution. A case study on uptake and release of PCB isomers and congeners in green-lipped mussels (Perna viridis) in Hong Kong waters. Environ Pollut. 1987;7:41–62.
11. Carvalho FP, Villeneuve JP, Cattini C, Rendon J, Oliveira M. Pesticide and PCB residues in the aquatic ecosystems of Laguna de Terminos, a protected area of the coast of Campeche, Mexico. Chemosphere. 2009;74:988–995.
12. Tolosa I, Mesa-Albernas M, Alonso-Hernadez CM. Organochlorine contamination (PCBs, DDTs, HCB, HCHs) in sediments from Cienfuegos Bay, Cuba. Mar Pollut Bull. 2010;60:1619–1624.
13. Liu Z, Zhang H, Tao M, et al. Organochlorine pesticides in consumer fish and mollusks of Liaoning Province, China: Distribution and human exposure implications. Arch Environ Contam Toxicol. 2010;59:444–453.
14. Mora P, Michel X, Narbonne JF. Cholinesterase activity as potential biomarker in two bivalves. Environ Toxicol Pharmacol. 1999;7:253–260.
15. Solé M, Porte C, Barcelo D, Albaiges J. Bivalves residue analysis for the assessment of coastal pollution in the Ebro Delta (NW Mediterranean). Mar Pollut Bull. 2000;40:746–753.
16. Viñas L, Franco A, Blanco X, et al. Temporal and spatial changes of PAH concentrations in Mytilus galloprovincialis from Ria de Vigo (NW Spain). Environ Sci Pollut Res. 2011;19:529–539.
17. O’Connor TP. Trends in chemical concentrations in mussels and oysters collected along the US coasts from 1986 to 1993. Mar Environ Res. 1996;41:183–200.
18. OSPAR. OSPAR Coordinated Environmental Monitoring Programme (CEMP). Agreement 2010–1
19. National Fisheries Research and Development Agency. An illustrated guide book on the mollusks from coasts of Korea. Busan: Guduck Publishing Co. (in Korean); 1999. p. 331.
20. Kim SK, Oh JR, Shim WJ, et al. Geographical distribution and accumulation features of organochlorine residues in bivalves from coastal areas of South Korea. Mar Pollut Bull. 2002;45:268–279.
21. Ramu K, Kajiwara N, Isobe T, et al. Spatial distribution and accumulation of brominated flame retardants, polychlorinated biphenyls and organochlorine pesticides in blue mussels (Mytilus edulis) from coastal waters of Korea. Environ Pollut. 2007;148:562–569.
22. Toba M, Natsume Y, Yamakawa H. Reproductive cycles of manila clams Ruditapes phillipinarum collected from Funabashi waters, Tokyo Bay. Nippon Suisan Gakkaishi. 1993;59:15–22.
23. Thompson S, Budzinski H, Garrigues P, Narbonne JF. Comparison of PCB and DDT distribution between water-column and sediment-dwelling bivalves in Arcachon Bay, France. Mar Pollut Bull. 1999;38:655–662.
24. Choi JY, Yang DB, Hong GH. Bioaccumulation of polychlorinated biphenyls (PCBs) and organochlorine pesticides in Manila clams (Ruditapes philippinarum) collected from the mid-western coast of Korea. Ocean Polar Res. 2010;32:237–245.
25. Bayne BL, Bubel A, Gabbott PA, Livingstone DR, Lowe DM, Moore MN. Glycogen utilisation and gametogenesis in Mytilus edulis L. Mar Biol Lett. 1982;3:89–105.
26. Gabbott PA. Developmental and seasonal metabolic activities in marine molluscs. Hochachka PW, editorThe Mollusca: Environmental Biochemistry and Physiology. 2:caNew York: Academic Press; 1983. p. 165.
27. Zhao Z, Zhang L, Wu J, Fan C. Distribution and bioaccumulation of organochlorine pesticides in surface sediments and benthic organisms from Taihu Lake, China. Chemosphere. 2009;77:1191–1198.
28. Yoo SH, Kim JB, Kim IK. Distribution aspect and seasonal distribution change of watering geese in Cheonsu Bay. Korean J Environ Ecol (in Korean). 2008;22:632–639.
29. Kwon SY, Lee JH, Hong JM, Hwang HB, Lee TW. Change in species composition of shallow water fish at the Namdang Beach after dike construction in Cheonsu Bay. Korean J Ichthyol (in Korean). 2013;25:106–118.
30. Park SW, Heo S, Yu J, et al. Temporal and spatial variation of water quality in the Cheonsu Bay of Yellow Sea, Korea. J Korean Soc Mar Environ Safety (in Korean). 2013;19:439–458.
31. Laurestein GG, Cantillo AY. Sampling and analytical methods of the national status and trends program, national benthic surveillance and mussel watch projects 1984–1992. NOAA Technical Memorandum NOS ORCA 71. 1993. p. 182.
32. Hedges JI, Stern JH. Carbon and nitrogen determinations of carbonate-containing solids. Limnol Oceanogr. 1984;29:657–663.
33. Qian Y, Serican JL, Wade TL. Determination of percent lipid in tissue. Lauenstein GG, Cantillo AY, editorsSampling and analytical methods of the National Status and Trends Program Mussel Watch Project: 1993–1996 Update. NOAA Technical Memorandum NOS ORCA 130. 1998. p. 27–29.
34. Ballschmite K, Zell M. Analysis of polychlorinated biphenyls (PCB) by glass capillary gas chromatography. Fresenius J Anal Chem. 1980;321:47–251.
35. KREI (Korean Rural Economic Institute) food balance sheet of Korea 2011 [Internet]. Korean Rural Economic Institute; c2010. Available from: http://krei.re.kr/kor/issue/report_view.php?Reportid=E05-2012&rclass=x&cpage=1
36. NRC (National Research Council), NAS (National Academy of Science). Risk assessment in the federal government: Managing the process. Washington D.C.: National Academy Press; 1983. p. 191.
37. US EPA. Risk based concentration table Integrated risk information system (IRIS), Philadelphia, PA. United States Environmental Protection Agency; Washington D.C.: 2004.
38. Howell NL, Suarez MP, Rifai HS, Koening L. Concentrations of polychlorinated biphenyls (PCBs) in water, sediment, and aquatic biota in the Houston Ship Channel, Texas. Chemosphere. 2008;70:593–606.
39. KARICO (Korea Agricultural & Rural Infrastructure Co.). Statistical yearbook of land and water development for agricultural 2000. Anyang: Korea Agricultural & Rural Infrastructure Co. (in Korean); 2000. p. 716.
40. Han KB, Park CK, Kim BY, et al. Agricultural environmental chemistry. Paju: DongHwa Technology Publishing Co. (in Korean); 1996. p. 430.
41. KARICO (Korea Agricultural & Rural Infrastructure Co.). Statistical year book of land and water development for agriculture 2002. Anyang: Korea Agricultural & Rural Infrastructure Co. (in Korean); 2002. p. 680.
42. Hong SH, Yim UH, Shim WJ, Li DH, Oh JR. Nationwide monitoring of polychlorinated biphenyls and organochlorine pesticides in sediments from coastal environment of Korea. Chemosphere. 2006;64:1479–1488.
43. Long ER, Morgan LG. The potential for biological effects of sediment-sorbed contaminants tested in the National Status and Trends Program. NOAA Technical Memorandum NOS OMA 52. National Oceanic and Atmospheric Administration; Seattle, Washington: 1990. p. 138.
44. Long E, Macdonald D, Smith S, Calder F. Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuarine sediments. Environ Manage. 1995;19:81–97.
45. Frignani M, Bellcci LG, Carraro C, Raccanello S. Polychlorinated biphenyls in sediments of the Venice Lagoon. Chemosphere. 2001;43:567–575.
46. Wurl O, Obbard JP. Organochlorine pesticides, polychlorinated biphenyls and polybrominated diphenyl ethers in Singapore coastal marine sediments. Chemosphere. 2005;58:925–933.
47. Iwata H, Tanabe S, Sakai N, Nishimura A, Tatsukawa R. Geographical distribution of persistent organochlorines in air, water and sediments from Asia and Oceania, and their implication for global redistribution from lower latitudes. Environ Pollut. 1994;85:15–33.
48. Hong SH, Yim UH, Shim WJ, Oh JR. Congener-specific survey for polychlorinated biphenyls in sediments of industrialized bays in Korea: Regional characteristics and pollution sources. Environ Sci Technol. 2005;39:7380–7388.
49. Hong SH, Yim UH, Shim WJ, Oh JR, Lee IS. Horizontal and vertical distribution of PCBs and chlorinated pesticides from Masan Bay, Korea. Mar Pollut Bull. 2003;46:244–253.
50. Zhang Z, Hong H, Zhou JL, Huang J, Yu G. Fate and assessment of persistent organic pollutants in water and sediment from Minjiang River estuary, Southwest China. Chemosphere. 2003;52:1423–1430.
51. Ma M, Feng Z, Guan C, Ma Y, Xu H, Li H. DDT, PAH, and PCB in sediment from the intertidal zone of the Bohai Sea and the Yellow Sea. Mar Pollut Bull. 2001;42:132–136.
52. Combi T, Taniguch S, Figueira LCR, de Mahiques MM, Martins CC. Spatial distribution and historical input of polychlorinated biphenyls (PCBs) and organochlorine pesticides (OCPs) in sediments from a subtropical estuary (Guaratuba Bay, SW Atlanti). Mar Pollut Bull. 2013;70:247–252.
53. Mohammed A, Peterman P, Echols K, et al. Polychlorinated biphenyls (PCBs) and organochlorine pesticides in harbor sediments from Sea Lots, Port-of-Spain, Trinidad and Tobago. Mar Pollut Bull. 2011;62:1324–1332.
54. Kucuksezgin F, Gonul T. Distribution and ecological risk of organochlorine pesticides and polychlorinated biphenyls in surficial sediment from the eastern Aegean. Mar Pollut Bull. 2012;64:2549–2555.
55. Barakat AO, Khairy M, Aukari I. Persistent organochlorine pesticides and PCB residues in surface sediments of Lake Qarun, a protect area of Egypt. Chemosphere. 2013;90:2467–2476.
56. Ballschmiter K, Wittlinger R. Interhemispheric exchange of hexachlorocyclohexanes, hexachlorobenzene, polychlorobiphenyls, and 1,1,1-trichloro-2,2-bis(p-chloro phenyl)-ethane in the lower troposphere. Environ Sci Technol. 1991;25:103–1111.
57. Phillips DJH. Trace metals and organochlorines in the coastal waters of Hong Kong. Mar Pollut Bull. 1989;20:19–327.
58. Villeneuve JP, Carvalho FP, Fowler SW, Cattini C. Levels and trends of PCBs, chlorinated pesticides and petroleum hydrocarbons in mussels from the NW Mediterranean coast: Comparison of concentrations in 1973/1974 and 1988/1989. Sci Total Environ. 1999;237/238:7–65.
59. Khaled A, El Nemr A, Said TO, El-Sikaily A, Abd-Alla AMA. Polychlorinated biphenyls and chlorinated pesticides in mussels from the Egyptian Red Sea coast. Chemosphere. 2004;54:1407–1412.
60. Carro N, Garcia I, Ignacio M, Mouuteira A. Organochlorine pesticides levels in Ensis siliqua (Linnaeus, 1758) from Ría de Vigo, Galicia (N.W. Spain): Influence of season, condition index and lipid content. Bull Environ Contam Toxicol. 2012;88:491–496.
61. Vieweg I, Hop H, Brey T, et al. Persistent orgainc pollutants in four bivalves species form Svalbard waters. Environ Pollut. 2012;161:34–142.
62. Li YF. Global gridded technical hexachlorocylohexane usage inventory using a global cropland as a surrogate. J Geophys Res. 1999;104:23785–23797.
63. Loganathan BG, Kannan K. Global organochlorine contamination trends: An overview. Ambio. 1994;23:187–191.
64. Won MS. Seed production and environmental influence on productivity of the short-necked clam, Ruditapes philippinarum. Ph D thesis. Busan: Busan National Fisheries Univ. (in Korean); 1994. p. 0–220.
65. Serrano R, Blanes MA, López FJ. Maternal transfer of organochlorine compounds to oocytes in wild and farmed gilthead sea bream (Sparus aurata). Chemosphere. 2008;70:16–566.
66. Oka M, Arai T, Shibata Y, Miyazaki N. Reproductive transfer of organochlorines in viviparous surf perch, Ditrema temmincki. Environ Pollut. 2006;142:383–387.
67. Park BK, Park GJ, An YR, Choi HG, Kim GB, Moon HB. Organohalogen contaminants in finless porpoises (Neophocaena phocaenoides) from Korean coastal waters: Contamination status, maternal transfer and ecotoxicological implications. Mar Pollut Bull. 2010;60:768–774.
68. Guiney PD, Melancon MJ, Lech JJ, Peterson RW. Effects of egg and sperm maturation and spawning on the distribution and elimination of polychlorinated biphenyl in rainbow trout (Salmo gairdneri). Toxicol Appl Pharmacol. 1979;47:261–272.
69. McManus GB, Wyman KD, Peterson WT, Wurster CF. Factors affecting the elimination of PCBs in the marine copepod Acartia tonsa. Estuar Coast Shelf Sci. 1983;17:421–430.
70. Hummel H, Bogaards RH, Nieuenhuize J, De Wolf L, Van Liere JM. Spatial and seasonal differences in the PCB content of the mussel Mytilus edulis. Sci Total Environ. 1990;92:155–163.
Fig. 1Sampling sites for sediments, Manila clams (Ruditapes philippinarum), and mussels (Mytilus coruscus) in Cheonsu Bay, Korea. 
Fig. 2a) PCB composition of commercial PCBs, Aroclor 1248 (A1248), −1254 (A1254), and −1260 (A1260), b) composition of PCB homologues, c) DDT metabolites, and d) HCH isomers in sediment samples from Cheonsu Bay. 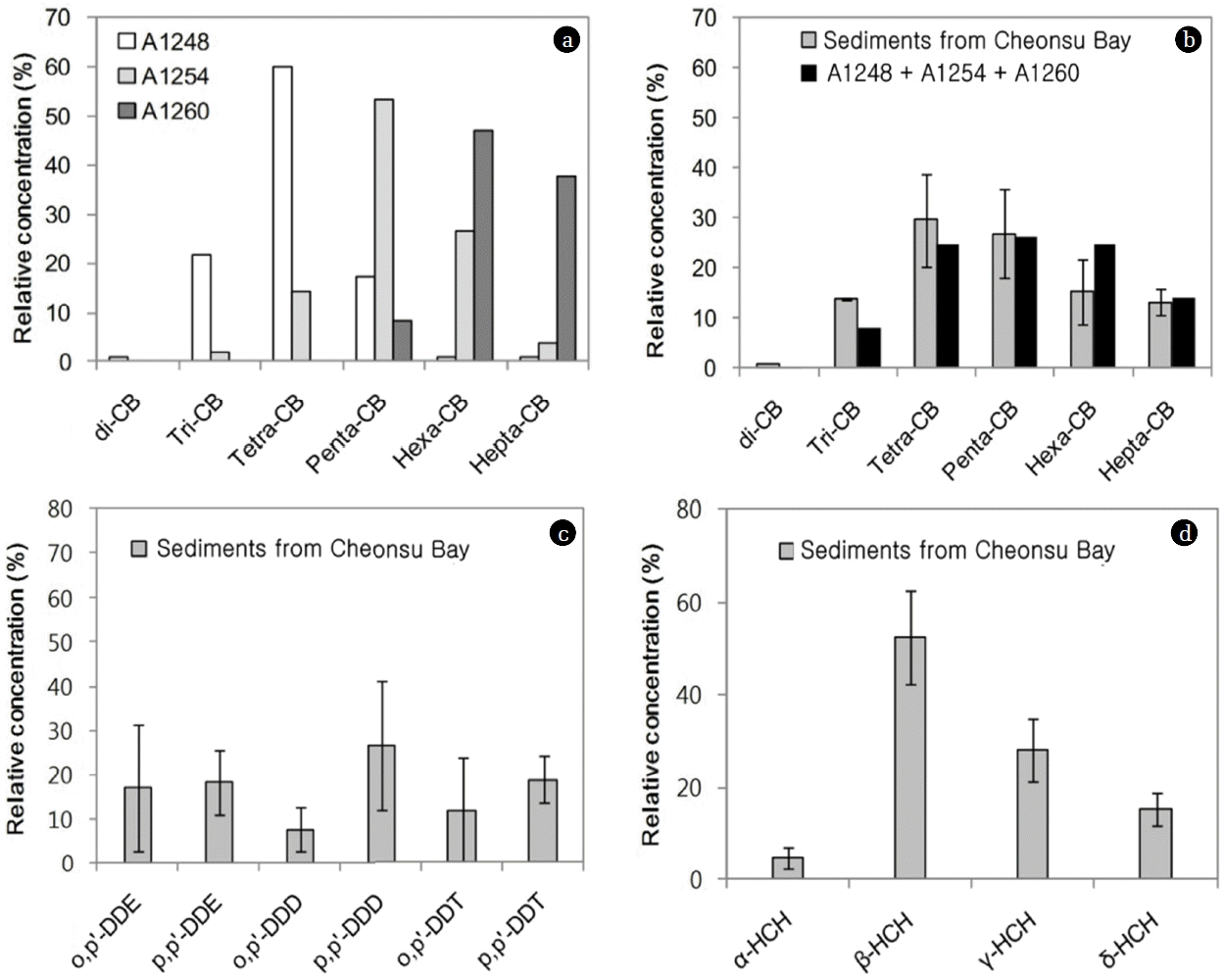
Fig. 3Seasonal changes in ∑PCBs, ∑DDTs, and ∑HCHs concentrations in Manila clams and mussels. Asterisks (*) indicate a significant difference value. One-way ANOVA: * p < 0.05. 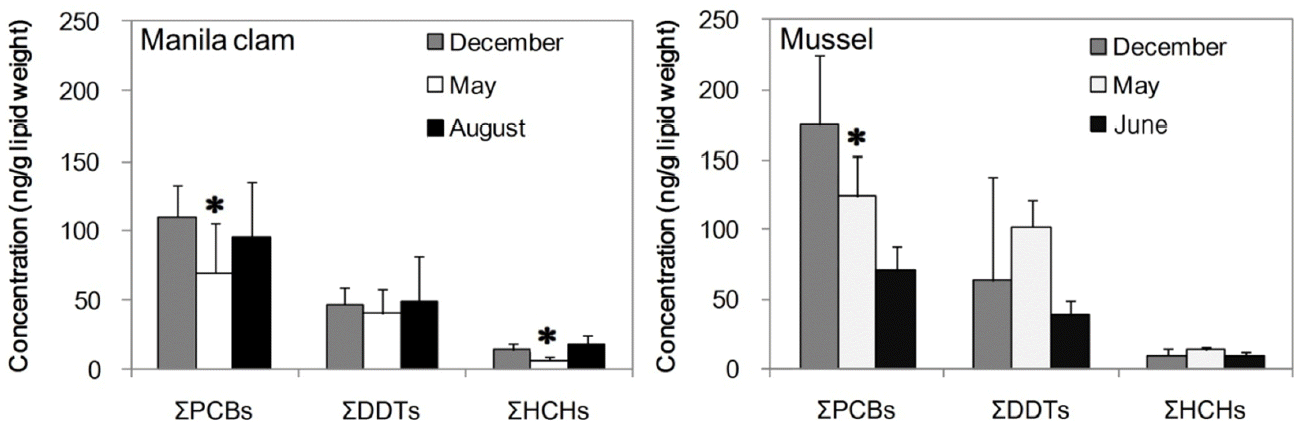
Fig. 4Dendrogram of hierarchical cluster analysis of important PCB congeners (PCB 44, 52, 87, 110, 138, and 153), p,p′-DDE, and β-HCH in sediment, Manila clam, and mussel samples. 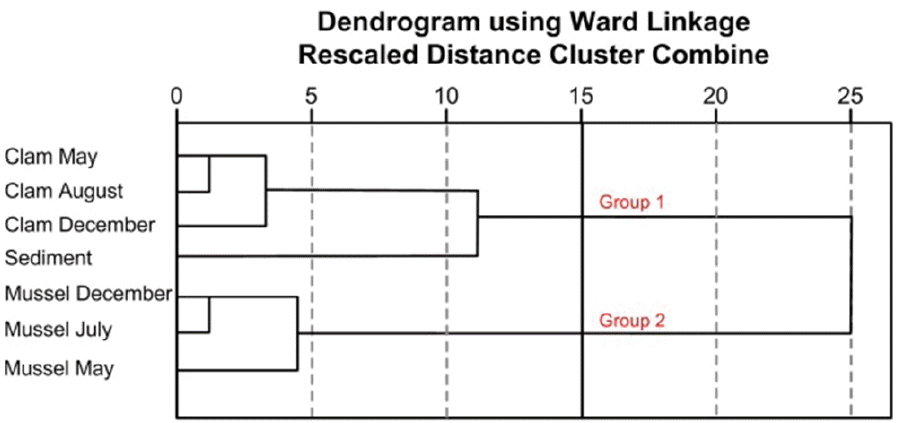
Table 1Mean Concentrations of ∑PCBs, ∑DDTs, and ∑HCHs in the Sediments Collected from St.1, 2, and 3 at the Cheonsu Bay, Korea, during the Sampling Period (Unit. ng/g dry weight) Total PCBsa estimates derived from the sum of 18 congeners multiplied by a standard factor of two [38], and < MDL is ‘below method detection limit’. Table 2The Levels of PCBs, DDTs and HCHs in a) Sediment and in the Literatures and Sediment Quality Guidelines Considered for Organochlorine Contaminants in Marine Sediments, and b) Bivalves Collected from Polluted Coastal Areas in References
Table 3Lipid Contents and Lipid-normalized Concentrations of PCBs and OCPs in Tissues of Manila Clams and Mussels Collected from Cheonsu Bay, Korea Table 4Life-time Cancer Risk (LCR) and Non-cancer Risk Posed by Consumption of Manila Clams and Mussels by Korean Population |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||