1. Introduction
Ecological risk assessment can be classified as probabilistic ecological risk assessment (PERA) and deterministic ecological risk assessment (DERA) according to quality and quantity of toxicity data [1]. PERA is based on species sensitivity distribution (SSD) and DERA uses the lowest toxicity value and assessment factor. Ecological risk assessment is used for effective and efficient environmental management. The environmental standard, guideline, or criteria was established based on risk assessment in developed countries including the Netherlands [2, 3], Australia [4], UK [5], United States [6], and Canada [7]. These countries have an autonomously administered risk assessment methodology for protection of native receptors. In the case of the Netherlands and Canada, the results from risk assessment, which is the lowest value both of human and ecological protective concentration (EPC), are used as an advisory value to set environmental standard, guidelines, or criteria. Presently, soil quality standard in Korea was based on human risk assessment. However, to protect environment there is a need to consider ecological risk assessment like the Netherlands and Canada. For Korean water environment management, EPC was derived by PERA [8, 9], and in the case of soil environment, the study of EPC based on risk assessment was not attempted. Meanwhile, An et al. [1] suggested ecological risk assessment methodology for soils in Korea by comparing methods of developed countries, such as the Netherlands [2], Canada [7], and European Commission (EC) [10].
For a case study to derive Korean soil EPC, cadmium (Cd) was selected because Cd was sorted to be given a priority as a toxic chemical [11] and soil contaminant [12]. It exists in nature as a nonessential heavy metal, but is hazardous. Cd is an identified carcinogen for humans by the International Agency for Research on Cancer (group 1) and the United States Environmental Protection Agency (B1: probable human carcinogen), and it is harmful to the ecosystem. In this study, we applied ecological risk assessment for Cd according to An et al. [1] and suggested the maximal permissible levels for soil EPC in Korea.
2. Materials and Methods
2.2. Selection of Data Based on Reliability
Collected toxicity data was assessed to be of grade 4 on the reliability criteria. Reliability 1 and 2 were used to derive soil EPC, whereas R3 and R4 were not used. R1 was based on Good Laboratory Practices and provided sufficient background information—test chemical, test process, test unit, moisture contents, and soil properties (e.g., texture, pH, and organic matter); R2 was based on Science Citation Index Expended papers, which provided sufficient background information; R3 did not provide sufficient background information; and R4 had only the toxicity values.
2.3. Classification of Acute and Chronic Data
According to An et al. [1], toxicity data was classified acute or chronic depending on the test duration. In the case of plants and earthworms, acute or chronic toxicity are separated by lesser than or greater than 8 weeks, respectively. In the case of Enchytraeid sp., six weeks was standard for assessing acute and chronic toxicity because it has a shorter duration for reproduction (4–6 weeks) than earthworm (8 weeks) [18]. Twenty-eight days are required for Folsomia candida reproduction and so a period less than 28 days is considered acute for the test duration. In case of Caenorhabditis elegans, less than 72 hr was considered acute. The endpoints of acute and chronic testing were expressed as effective/lethal concentration 50 (EC50/LC50) and no/lowest observed effect concentration (NOEC/LOEC), respectively.
2.4. Normalization to Standard Soil
Toxicity test was performed in various soils. In case of natural soil, their property was different based on the location. Organisation for Economic Co-operation and Development (OECD) artificial soil was composed of 70% sand, 10% peat moss, and 20% kaolin. The soil properties such as organic matter and clay contents were related to toxicity results. In EC, UK, the Netherlands, and Australia, the toxicity data was normalized in consideration to the soil properties.
2.5. Deriving Soil EPC
Soil EPC was calculated using a domestic soil ecological risk assessment methodology [1]. The methodology is distinct as PERA and DERA depend on quantity and quality of toxicity data. PERA (e.g., statistical extrapolation method [SEM] and SEM acute to chronic ratio [SEMACR]) is based on SSD. SEM is applied to sufficient data for chronic N(L)OEC or acute E(L)C50 for more than 4 taxonomic groups including plants (Magnoliopsida or Liliopsida), earthworm (Clitellata), and macroorganism (Insecta or Secernentea). SEM is calculated from 5 percentile and 50 percentile hazard concentrations (HC5 and HC50) of SSD by ETX program, which was developed by the Rijksinstituut voor Volksgezondheid en Milieu (RIVM; National Institute of Public Health and Environment). The HC5 and HC50 were calculated as following equation [19];
where, HCp is hazardous concentration for p% of species; χ̄ is mean of log transformed NOEC value; k is extrapolation factor [19]; s is standard deviation of log transformed toxicity data. HC5 and HC50 are automatically calculated as above equation with 90 % confidence interval by the ETX program.
For consideration to land use, HC5 and HC50 apply to residential/agricultural and industrial/commercial land, respectively. SEMACR is applied when chronic data was insufficient but acute data was sufficient. At this time, acute HC5 and HC50 divide the ACR, which is ratio of geometric mean of acute and chronic values of the same species. If it is impossible to calculate ACR or is less than 10, then the ACR is considered with 10 as default value. To consider uncertainty of derived HC5 (or HC50), assessment factor for SSD was applied. In Korea, we do not have standard soil properties yet. In this study, toxicity data was not normalized to Korean standard soil. Therefore, HC5 (or HC50) was divided by 5. DERA, which is based on an assessment factor method, is obtained from the lowest toxicity values (for residential/agricultural land) or geometric mean of toxicity (for industrial/commercial land) values divided by assessment factor as land use. Finally, the soil EPC is sum of HC5 (or HC50) and background concentration for Cd.
3. Results and Discussion
All the soil toxicity data collected for Cd was confirmed by previous references and the reliability of the data was assessed. Soil toxicity data, which belonged to R1 and R2, was listed in Table 1 and 2 In the case of acute toxicity, 5 classes of toxicity data (2 Magnoliopsida, 6 Liliopsida, 17 Clitellata, 3 Insecta, and 5 Secernentea) were sorted (Table 1). Plant acute toxicity data was included for Zea mays, Sorghum bicolor, Triticum aestivum, and Cucumis sativus. Earthworm acute toxicity data was determined using Eisenia fetida and Enchytraeus albidus and acute toxicity data of Folsomia candida and Caenorhabditis elegans were collected. In the case of chronic toxicity, 4 classes of toxicity data (1 Liliopsida, 7 Clitellata, 28 Insecta, and 2 Secernentea) were sorted (Table 2). Chronic data from one species of plant (Andropogon scoparius), chronic data from three species of earthworm (Eisenia fetida, Eisenia andrei, and Lumbricus rubellus) were used in the derivation of EPC. In addition, Folsomia cadida, Proisotoma minuta (Insecta), and Plectus communis (Secernentea) were collected.
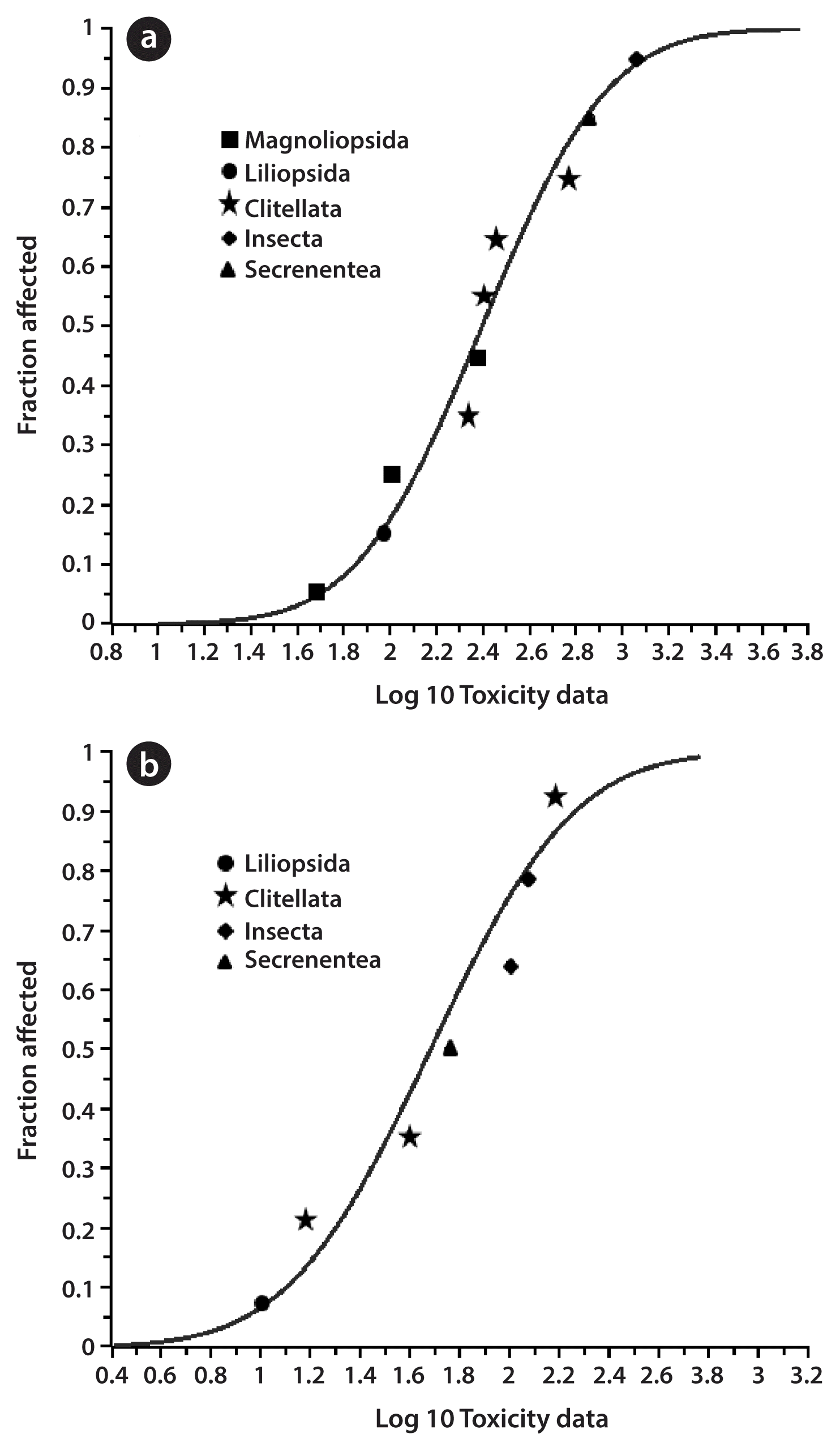
Both the acute and chronic data were collected for more than 4 samples, and therefore, soil EPC can be applied using PERA. The input data for SSD that was used was the species mean acute value or species mean chronic value, which was calculated based on the geometric mean of the same species (Fig. 1). The acute HC5 and HC50 were calculated as 46.93 (14.35–91.73) and 248.47 (140.83–438.38) mg/kg, respectively. The chronic HC5 and HC50 were 7.89 (1.39–18.39) and 47.98 (22.32–103.15) mg/kg. Rationally, the toxicity data was to be normalized to Korean standard soil; however, studies of Korean standard soil properties were not done yet. Therefore, assessment factor of 5 was applied. The background concentration of Cd was 0.25 mg/kg [20]. The final acute and chronic EPC for residential/agricultural land use was 9.39 and 1.58 mg/kg, respectively. The final acute and chronic EPC for industrial/commercial land use was 49.69 and 9.60 mg/kg, respectively.
The soil EPC, which is called as predicted-no-effect-concentration (PNEC) by the EC, soil screening value (SSV) in UK, maximum permissible concentration (MPC) in the Netherlands, soil quality guidelines (SQG) in Canada, and ecological soil screening level (Eco-SSL) in Unite States, of Cd is already reported in EC, UK, the Netherland, Canada, and United States. In European countries, such as UK and the Netherlands, EPC was based on HC5. In EC, they applied SEM method based on the data quality groups from R1 (according to standard test) to R3 (no standard procedure and missing some background information, such as pH, measured concentration of chemicals, statistical results, origin of test species, the range of test concentrations). HC5 of Cd was derived from 21 NOEC data of microflora (R1–R3) by ETX program and it was calculated to be 2.3 mg/kg. Finally, PNEC of Cd was reported as 1.15–2.3 mg/kg, after application of assessment factor of 1–2 because of uncertainty of data [10]. In the Netherlands, maximum permissible addition, which was based on HC5 and was not on the added background concentration, was derived to be 0.79 mg/kg [21]. This value was based on 13 NOECs for 6 taxonomic groups including 3 macrophyta, 1 mollusca, 5 annelida, 1 isopoda, 2 insecta, and 1 arachnida. Serious risk addition, which was based on HC50, was derived as 12 mg/kg [21]. The SSV of UK was 1.5 mg/kg that equates to the EC standards [5]. In the case of Canada, SQG for agricultural, residential/parkland, commercial, and industrial land were 3.8, 10, 22, and 22 mg/kg [22]. The Eco-SSL of United States was derived according to receptors involved. The Eco-SSL for plant and soil invertebrates was 32 and 140 mg/kg, respectively [14]. Eco-SSL for plant was calculated from 14 plant species of maximum acceptable toxicant concentration (MATC) values. Eco-SSL for invertebrate was derived from the MATC and EC10 of 3 invertebrate species, such as Eisenia andrei, Folsomia candida, and Plectus acuminatus. Since, Eco-SSL was calculated depending upon the geometric mean of toxicity values for plant or soil invertebrates, these values were higher than other countries. Generally, for the establishment of EPC chronic values of any pollutant are used. Data set and methodology for soil EPC are different with other country. However, soil EPC in Korea was similar with EPC of EC, the Netherlands, UK, and Canada. This reason may be that these countries calculate EPC based on SSD. Meanwhile, the Korean soil standard for residential/agricultural and industrial/commercial land were 4 (1 region), 10 (2 region), and 60 (3 region) mg/kg. These values are higher than EPC because the soil standard was based on human risk.
4. Conclusions
In present study, we derived soil EPC of Cd in Korea using PERA based on SSD. The soil EPC was calculated according to land use for residential/agricultural and industrial/commercial purposes. As the results, the chronic soil EPCs, which were generally used for EPC in other counties, for residential/agricultural and industrial/commercial lands were derived to be 1.58 and 9.60 mg/kg, respectively. Though these values were similar to EPCs of EC, the Netherlands, UK, and Canada, soil EPCs of Cd were lower than the Koreas soil standard, which was currently established based on human risk. Therefore, there is a need to consider the ecological effects of soil pollutants in the near future.