AbstractUsing the abandoned agricultural by-product corncobs, the most commonly used methylene blue (MB) dyestuffs were removed. This experiment is very meaningful because it is the recycling of resources and the use of environmentally friendly adsorbents. According to the Hauser ratio and porosity analysis, the corncob has a good flow ability of the adsorbent material and many pores, which is very advantageous for MB adsorption. As a result of the experiment, MB concentration of less than 0.005 g/L was very efficiently removed with 10 g/L of bioadsorbent corncob and the maximum adsorption capacity of corncob for MB dyes was obtained at 417.1 mg/g. In addition, adsorption process of MB onto corncob was a physical process according to adsorption energy analysis. Corncob can efficiently and environmentally remove MB in aqueous solution, and is very cost effective and can recycle the abandoned resources.
1. IntroductionDyes are used in many industries closely related to human life such as textiles, leather products, paper, and plastics [1]. Today, more than 10,000 dyes are commercially available. The amount of dye wastewater generated annually worldwide is estimated to be about 750,000 tons [2]. The number of textile companies in Korea related to dyeing is 360,616 in 2015, equivalent to 3.6% of total wastewater discharging facilities. However, dyeing wastewater emission accounts for 22.2% of total wastewater emission. Moreover, the organic matter load from dyeing wastewater is 2,134,429 kg/d, corresponding to 24.3% of total wastewater discharge [3, 4]. For this reason, dyeing wastewater is classified as a representative pollution from industry. Most dyes have complex and stable structures such as aromatic structures. Therefore, they are difficult to be decomposed by chemical, physical, or biological treatment [5]. When these dyestuffs are discharged without treatment, chromaticity can cause aesthetic discomfort. In addition, the dye can interfere with sunlight penetration of water surface, thereby deteriorating photochemical and biological activities of aquatic plants [2]. Dyes also have the potential to cause groundwater contamination, posing potential threat to assimilation of organisms [4, 6]. Methylene blue (MB) is one inexpensive cationic dye widely used for dyeing cotton, wool, silk, leather, and coating paper [7]. Among various dyes classified, cationic dyes are known to be more toxic than other dyes. Although MB is not more toxic compared to other cationic dyes, long-term exposure to MB can cause serious health problems such as increased heart rate, vomiting, shock, cyanosis, jaundice, limb paralysis, mental confusion, eye burns, and tissue necrosis [8]. To reduce the impact of MB on the environment, animals, and plants, MB contaminated wastewater should be treated before being discharged into water environment.
Currently, dye wastewater is treated by various methods such as ozone oxidation treatment, coagulation treatment, advanced oxidation (AOP), and biological treatment [9]. The coagulation process is effective in removing insoluble dyes such as disperse dyes. However, it is ineffective for soluble dyes with disadvantage of producing large amounts of sludge [10]. The ozone treatment process is effective for removing almost all dyes except disperse dyes. However, ozone is costly because it has a short half-life. It needs to be supplied continuously for dye removal [11]. Most known dye removal processes have low removal rates. Particularly, they have very low removal efficiencies for samples with low dye concentrations. On the other hand, the adsorption method is simple to operate and has high removal efficiency. Thus, it is widely used in the treatment of dye wastewater [12, 13]. However, activated carbon is expensive. Moreover, it needs additive materials to improve removal rate of inorganic substance [14].
In recent years, many researches have been actively carried out to remove dyeing wastewater and heavy metals using agricultural byproducts such as wheat straw [15], banana peel [16], pomelo peel [17], bagasse [18], rice husks [19], tea wastes [7], canola residues [10], cashew nut shell [20, 21], tamarind seed [22], and orange peel [23]. These studies have mainly used adsorbents by activating low-cost agricultural wastes or by-products. However, MB removal rate was low due to diffusion limitation and decreased surface active sites. Further, there is problem in the separation and removal of adsorbent after wastewater treatment and generation of secondary wastes. Therefore, it is necessary to develop an ecofriendly adsorbent that is inexpensive with increased adsorption for MB. In order to overcome these problems, corncob was used as a low-cost adsorbent to remove MB dye in this study.
Maize (Zea mays) is the world’s third largest food crop after rice and wheat. Global corn production is estimated at over 1,016,431,000 tons per year in 2015 [24]. The amount of corncob wastes is increasing with increasing corn production. Corncob is one of the most agricultural wastes produced in the world. According to a previous report [25], 18 kg corncobs are generated when 100 kg of corn grains are produced. Therefore, the aim of this study was determine the adsorption rate of corncob for MB and compare the result with the adsorption rate reported by other researchers. In addition, adsorption kinetics and intraparticle diffusion were analyzed by applying pseudo-first-order and pseudo-second-order. Moreover, adsorption isotherms of Langmuir, Freundlich, and Dubinin-Radushkevich were investigated and thermodynamic analyses were carried out by adsorption experiments through temperature changes.
2. Materials and MethodsAgricultural waste adsorbent, corncobs, were collected from farms in Gangneung, Korea. After collection, corncobs were washed several times with deionized water to remove contaminants on the surface of corncob followed by drying in an oven at 70°C for 72 h. Dried corncobs were cut into 0.5 cm in thickness and stored in a desiccator for experiments. MB (C16H18N3SCI, Sigma Aldrich) is cationic dye and heterocyclic aromatic chemical compound.
In this experiment, stock solution of MB was prepared by dissolving 0.02 g of MB in 1 L of distilled water. Depending on the concentration of MB required for the experiment, it was diluted. Batch studies were conducted to investigate the effect of pH, initial MB concentration, temperature, and initial corncob dosage on adsorption efficiency for MB. pH and temperature were controlled from 2 to 10 using HCl and/or NaOH solution and from 10°C to 30°C using electrical thermostat, respectively, to effect adsorption of MB onto corncob. Different amounts of corncobs and 500 mL of MB solution were then added into a 1 L of conical flask and stirred with a magnetic bar at 200 rpm. Contents of these flasks were sampled at fixed intervals of time (0, 10, 20, 30, 60, 120, 240, 720, and 1,440 min) and filtered through membrane filter (0.45 μm, Whatman). Concentrations of MB dye remained in these filtered samples were analyzed using spectrophotometric method. All experiments were carried out by changing one parameter at a time while holding the others constant. Experiments were performed five times. Mean values and standard deviation were calculated.
Qualitative and quantitative analyses of inorganic components contained in corncob were performed using X-ray fluorescence (XRF) (XRF-1500, Shimadzu, Japan). Surface analysis was performed using Scanning electron microscope (SEM, SM-300) and Topcon, respectively. The measurements of ash content, porosity, particle density and Hausner ratio were in accordance with the measurement method published by Azubuike and Okhamafe [26]. The size of the corncob particles was measured using a particle size analyzer (Laser Diffraction Master class 3 & 4, Malvern, England). The amount of corncob was measured with an electronic balance (XP26, Mettler Toledo, Swiss). The pH and temperature were measured using a pH meter (SevenGO pro, Mettler Toledo).
The adsorbed amount of MB by corncob was measured using UV-vis spectrophotometer (Shimadzu, MINI-1240) at 664 nm, the maximum absorption wavelength of MB. The adsorption amount was calculated using the following equation:
where Co (mg/L) was the initial concentration of solution, Ct (mg/L) was the concentration of solution at time t, V (L) was the volume of solution, and m (g) was the amount of corncob.
Percentage removal of MB dye was calculated using the following equation:
where C0 and CF were the initial and final concentration of MB dye in the solution, respectively.
3. Results and Discussion3.1. Characteristics of CorncobRegarding the content of dried corncob, there were 86.4 g of carbohydrate and 0.8 g of fat. Results of general composition analysis of the dried corncob are shown in Table 1. Fixed carbon represents the percentage of carbon remaining after subtracting the proportion of moisture, ash, and volatiles. Total carbon content of dried corncob was 58% and fixed carbon content was 38%. Ash content plays an important role in the adsorption of electrolyte from solution when it is present in small amounts [26]. Results showed that the ash content of dried corncob was as low as 3%. Contents of sulfur and nitrogen were also low as 0.03% and 0.83%, respectively. Therefore, nitrogen and sulfur based functional groups are not expected to affect the adsorption of MB by corncob. The Hausner ratio can be used to estimate cohesion between particles. A Hausner ratio of less than 1.20 indicates good flow ability, and a value higher than 1.50 indicates poor flow properties [26]. The Hausner ratio of corncob in this study was determined 0.893, indicating good flow ability of the adsorbent material. The porosity of the adsorbent is another criterion for selecting good adsorbents. The porosity of the corncob used in this experiment was measured around 83.5%, indicating very high porosity, which is expected to have a good effect on adsorption of MB.
SEM image of corncob is shown in Fig. 1. The surface of corncob was very irregular and porous in nature (Fig. 1). These porous and irregular surfaces are thought to be very helpful for adsorbing MB dye [24]. In addition, the surface of the corncob was composed of multiple layers of thin films. This can be useful to adsorb heavy metals and dyes [13].
3.2. Effect of Different Parameters on MB Removal3.2.1. Effect of initial MB concentrationThe initial MB dye concentration affords an important driving force in overcoming mass transfer resistance between the corncob surface and MB dye solution. The effect of initial dye concentration on the removal of MB dye by corncob is shown in Fig. 2(a). The adsorption rate, adsorption capacity, and the cost of the adsorbent are very important factors in the selection of adsorbent. The amount of adsorbent is also important in terms of cost. The ability to adsorb and remove large quantities of harmful substances with a small amount of adsorbent has many economic advantages. Therefore, in this experiment, we investigated the concentration of MB that could be removed by corncob adsorbent. Corncob 10 g/L was added to various concentrations of MB (0.02 g/L, 0.01 g/L, 0.005 g/L, and 0.0025 g/L) in solution. The effect of MB concentration on its removal rate was observed for 24 h. In 3 h, corncob removed 82.46%, 86.79%, 96.01%, and 98.62% of MB at MB concentrations of 0.02 g/L, 0.01 g/L, 0.005 g/L, and 0.0025 g/L in aqueous solution, respectively (Fig. 2(a)). The percentage removal of MB dye was decreased gradually with an increase of MB dye concentration from 0.0025 to 0.02 g/L. This is because the capacity of corncob capable of adsorbing MB is sufficient for low concentration of MB dye. This could decrease the adsorption capacity of corncob at higher concentration of MB dye. Therefore, the removal percentage of MB was decreased when MB dye concentration was increased.
After 3 h, the removal rate of MB by corncob was not significantly changed. Adsorption equilibrium was reached in 5 h. After 24 h, the percentage of MB adsorbed onto corncob was found to be 84.05%, 91.48%, 99.90%, and 99.97% for 0.02 g/L, 0.01 g/L, 0.005 g/L, and 0.0025 g/L of MB, respectively. According to experimental results, MB concentration below 0.005 g/L could be removed almost 100% with 10 g/L corncob.
3.2.2. Effect of pHpH, alkalinity, stirring conditions, and static materials are main influential factors in the adsorption and removal of oil and inorganic materials in aqueous solution. In particular, temperature and pH of aqueous solution can greatly influence the removal of dyes [27]. To investigate the effect of pH on MB removal rate, pH was controlled at 2, 4, 6, 8, and 10. The removal rate of MB by corncob was 11.47% at pH 2, 38.46% at pH 4, 99.74% at pH 6, 99.89% at pH 8, and 99.90% at pH 10 (Fig. 2(b)). The adsorption of MB using corncob depended strongly on the pH of the aqueous solution. This is because the pH of the solution is affected by dissolved state of MB in aqueous solution and surface charge distribution of the corncob. That is, the pH can affect the electrostatic interaction of the type of MB in the aqueous solution and the rebound characteristics between surfaces of the corncob. MB is present in aqueous solution as cationic species as undissociated molecules. At pH 3, undissociated molecules of MB predominate at 86%, with 50% of cationic species and undissociated molecules at pH 4. Only cationic species of MB are present at pH 6 or above [28]. That is, the higher removal rate at higher pH is due to increased anion in the aqueous solution as pH increases, making it easier to adsorb MB present as cationic species.
3.3. Adsorption Kinetics by CorncobThe kinetic mechanism for the adsorption of MB dye by corncob plays an important role in the adsorption process. It provides important information about the reaction pathway and the rate-controlling mechanism of exchange reactions [10]. In the present study, pseudo-first-order, pseudo-second-order, and intra-particle diffusion were used to examine the adsorption and diffusion rates of MB by corncob. Pseudo-first-order reaction depends only on the concentration of one reactant (a single-molecule reaction). Other reactants might be present. However, each will be in zero order. Pseudo-second-order reaction depends on the concentration of one second order reactant or two first order reactants. The calculated experiment data are shown in Fig. 3(a) and (b), and adsorption kinetics parameters are shown in Table 2. Among all models, calculated adsorption capacity values of pseudo-second-order models were the closet to experimental adsorption capacity. In addition, the coefficient of determination (R2) was found to be 0.8327 for pseudo-first-order model and 0.9968 for pseudo-second-order model when adsorption data of MB by corncob were applied. Therefore, the pseudo-second-order kinetic model had a better fit for experimental data compared to pseudo-first-order kinetic model considering its higher correlation coefficient value. This suggests that adsorption process is controlled by sorption between molecules of MB dye and the surface of corncob. Moreover, results showed that the rate of adsorption depended on the availability of adsorption sites on the surface of corncob rather than the number of adsorbed MB dye molecules. Fig. 3(c) shows the result when the internal particle diffusion model of equation is applied. It shows that data do not have a linear relationship. These results suggest that the adsorption process of MB dye on corncob occurs with complex mechanisms involving external mass transfer and internal particle diffusion. As shown in Fig. 3(c), mass transfer occurs in the adsorption initial region (linear region of t1/2 < 1.7). Then it occurs in subsequent linear region (linear region of t1/2 > 1.7) as an adsorption process involving internal particle diffusion. The intra-particle diffusion rate constant of MB obtained from the slope of equation was at 53.56 mg/g/h0.5.
3.4. Adsorption IsothermInteraction between adsorbate and adsorbent surface, maximum adsorption capacity of the adsorbent material, and dynamic equilibrium of adsorption system were investigated using the adsorption isotherm model. It has been reported that the best fitted adsorption isotherm model can provide information about the nature of the adsorption system [2]. In this adsorption study, three parameter models (Langmuir, Freundlich model, and Dubinin-Radushkevich) were used to fit with experimental data to estimate the best fitted isotherm model. Results of applying equations to data obtained from equilibrium adsorption experiments are shown in Fig. 4(a), (b) and (c). Langmuir, Freundlich, and Dubinin-Radushkevich isotherm constants for the adsorption of MB by corncob are shown in Table 3 According to the value of R2, the adsorption isotherm study indicated that the Langmuir isotherm model fitted better than the Freundlich isotherm model. This indicates that the adsorption process is more monolayer in nature [29]. The maximum adsorption of MB from the Langmuir equation was found to be 417.12 mg/g. The average adsorption energy value obtained from the Dubinin-Radushkevich isotherm model indicated physical adsorption or chemical adsorption of MB by corncobs. In general, the adsorption energy is 1 to 8 kJ/mol for physical adsorption and 20 to 40 kJ/mol for chemical adsorption [30]. In this study, the adsorption energy of MB was found to be 3.352 kJ/mol (Table 2). Therefore, the adsorption process of MB by corncob is more influenced by physical adsorption. Moreover, the Freundlich exponent n can be used to describe the intensity of adsorption. In this experiment, the value of n was measured to be 3.108. Therefore, the MB adsorption process was closer to a physical process. According to general interpretation of n and 1/n value, n = 1 indicates linear, n > 1 indicates physical process, n < 1 indicates chemical process, 1/n = 0.1 to 0.5 indicates that adsorption process is magnificent, 1/n = 0.5 to 1 indicates that the adsorption process is easy, and 1/n ≥ 1 indicates that the adsorption process is difficult [30]. In this experiment, the value of 1/n was found to be 0.32, indicating that the MB dye adsorption process by corncob was very favorable and magnificent.
Results of comparison of the maximum adsorption capacity of MB dye by agricultural byproduct adsorbent are shown in Table 3. The maximum adsorption capacity of MB in the case of corncob used as an adsorbent in this study showed a higher adsorption amount than those of others. When agricultural byproducts were modified to remove the dyes by adsorption, removal efficiencies were relatively high than those of natural materials.
3.5. Thermodynamic InterpretationAdsorption isotherm is affected by temperature. It represents thermodynamic equilibrium between MB adsorbed on the surface of corncob and MB in the aqueous solution. The effect of temperature on the capacity of corncob to adsorb MB was analyzed by adsorption isotherms of MB by corncob from aqueous solution at 20, 30, and 40°C with pH at 6 (Fig. 5(a)). Adsorption capacities were found to be 423, 452, and 490 mg/g at 20, 30 and 40°C, respectively. This tendency indicated that the adsorption capacity was increased by 1.07 and 1.16 times when the temperature was increased from 20°C to 30°C and from 30°C to 40°C, respectively.
In order to investigate the effect of temperature change on adsorption capacity, equilibrium adsorption experiments were carried out at various solution temperatures to 293K, 303K, and 313K. The value of ΔH° and ΔG° were calculated from slope and intercept of the plot of InkL versus 1/T. Change in Gibbs free energy was calculated by using equation. Results are shown in Fig. 5(b). Calculated thermodynamic parameters for the removal of MB by corncob are shown in Table 2. Gibbs free energy is −20 to 0 kJ/mol for physical adsorption and −80 to −400 kJ/mol for chemical adsorption [30]. In adsorption experiments of this study, the value of ΔG° was about −13.41 to −16.58 kJ/mol, which fell in the range of physical adsorption process. In addition, ΔG° was negative, indicating that the adsorption process was spontaneous in nature. The positive value of ΔH° at all temperature indicated that the adsorption of MB dye by corncob was endothermic in nature. This might be due to the strong adsorptive force between the surface of the corncob and the MB dye molecule. In endothermic process, the adsorption equilibrium can be increased by raising the temperature. The value of ΔH°, an important point in physical or chemical adsorption process, was measured to be 18.12 kJ/mol. Generally, if ΔH° value is less than 40, it is interpreted as physical adsorption [21]. In addition, the affinity of MB dye for corncob is good because ΔS° is positive at 0.1057 J/mol·K. The positive value of ΔS° indicates that MB dye concentration is increased in the solid-liquid interface, thereby decreasing MB dye concentration on the surface of corncob. Moreover, the positive value of ΔS° might be featured that adsorbate molecules gets at least one degree of freedom when it loses adsorption on the corncob. It also validates that randomness at the solid-liquid interface is increased during the adsorption process. Taking all results into consideration, MB removal using corncob is more likely to be performed by physical adsorption using the force of van der Waals rather than by chemical adsorption.
4. ConclusionsBatch adsorption experiments were carried out to remove MB in water by using corncob, an agricultural waste, as an adsorbent to optimize the influence of parameters such as initial MB dye concentration, pH, adsorbent dosage, and temperature on MB removal. Corncob bioadsorbent successfully removed MB dye in the aqueous solution (up to 96% of MB) with the following optimal parameters: Temperature 30°C; pH > 6; corncob 10 g/L; and MB concentration < 0.005 g/L. These results indicate that corncob can be a promising bioadsorbent due to its high efficiency and low dose requirements. The maximum adsorption of MB from the Langmuir equation was found to be 417.1 mg/g. The adsorption kinetic study was best described by pseudo-second-order kinetic model. This indicates that the adsorption process is controlled by physical adsorption. In addition, the value of 1/n was 0.32. This indicates that the MB dye adsorption process onto corncob is very favorable and magnificent. In the adsorption process, external mass transfer occurs in the early stage. Internal particle diffusion process then proceeds. The negative ΔG° value indicates that the adsorption is feasible and spontaneous. The positive ΔH° and ΔS° values indicate the endothermic nature of the adsorption and the affinity of corncob for MB adsorption, respectively. Based on these experimental results, we conclude that corncob is a cheap, efficient, and commercially viable new eco-friendly adsorbent for adsorbing and removing MB dye.
AcknowledgmentsThis study was supported by a grant (2016005271) of the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology, Republic of Korea.
References1. Annadurai G, Juang RS, Lee DJ. Use of cellulose-based wastes for adsorption of dyes from aqueous solutions. J Hazard Mater. 2002;B92:263–274.
2. Ahmed MJ, Dhedan SK. Equilibrium isotherms and kinetics modeling of methylene blue adsorption on agricultural wastes-based activated carbons. Fluid Phase Equilib. 2010;317:9–14.
3. Lee S, Ong S. Oxalic acid modified rice hull as a sorbent for methylene blue removal. APCBEE Procedia. 2014;9:165–169.
4. Choi HJ, Kim KH. Parametric study a dyeing wastewater treatment by modified sericite. Environ Technol. 2016;37:2572–2579.
5. Peydayesh M, Kelishami AR. Adsorption of methylene blue onto Platanus orientalis leaf powder: Kinetic, equilibrium and thermodynamic studies. J Ind Eng Chem. 2015;21:1014–1019.
6. Yagub MT, Sen TK, Afroze S, Ang HM. Dye and its removal from aqueous solution by adsorption: A review. Adv Colloid Interf Sci. 2014;209:172–184.
7. Ebrahimian A, Saberikhah E, Badrouh M, Emami MS. Alkali treated foumanat tea waste as an efficient adsorbent for methylene blue adsorption from aqueous solution. Water Resour Ind. 2014;6:64–80.
8. Gong R, Li M, Yang C, Sun Y, Chen J. Removal of cationic dyes from aqueous solution by adsorption on peanut hull. J Hazard Mater. 2005;B121:247–250.
9. Rafatullah M, Sulaiman O, Hashim R, Ahmad A. Adsorption of methylene blue on low-cost adsorbents: A review. J Hazard Mater. 2010;177:70–80.
10. Balarak D, Jaafari J, Hassani G, et al. The use of low cost adsorbent (Canola residues) for the adsorption of methylene blue from aqueous solution: Isotherm, kinetic and thermodynamic studies. Colloid Interf Sci Commun. 2015;7:16–19.
11. Kamel HA, Jibouri A, Wu J, Upreti SR. Continuous ozonation of methylene blue in water. J Water Process Eng. 2015;8:142–150.
12. Kumar PS, Abhinaya RV, Arthi V, Gayathrilashmi K, Priyadharshini M, Sivanesan S. Adsorption of methylene blue dye onto surface modified cashew nut shell. Environ Eng Manag J. 2014;13:545–556.
13. Sych NV, Trofymenko SJ, Poddubnaya OI, et al. Porous structure and surface chemistry of phosphoric acid activated carbon from corncob. Appl Surf Sci. 2012;261:75–82.
14. Abdelfattah I, Ismail AA, Sayed FA, Almedolab A, Aboeloghait KM. Biosorption of heavy metals ions in real industrial wastewater using peanut husk as efficient and cost effective adsorbent. Environ Nanotechnol Monit Manag. 2016;6:176–183.
15. Pirbazari AE, Saberikhah E. Fe3O4– wheat straw: Preparation, characterization and its application for methylene blue adsorption. Water Res Ind. 2014;7–8:23–37.
16. Pereira H, Carvalho D, Huang J, et al. Improvement of methylene blue removal by electrocoagulation/banana peel adsorption coupling in a batch system. Alexandria Eng J. 2015;54:777–786.
17. Argun ME, Guclu D, Karatas M. Adsorption of reactive blue 114 dye by using a new adsorbent: pomelo peel. J Ind Eng Chem. 2014;20:1079–1084.
18. Low LW, Teng TT, Morad N, Azahari B. Studies on the adsorption of methylene blue dye from aqueous solution onto low-cost tartaric acid treated bagasse. APCBEE Procedia. 2012;1:103–109.
19. Vadivelan V, Kumar KV. Equilibrium, kinetics, mechanism, and process design for the sorption of methylene blue onto rice husk. J Colloid Interf Sci. 2005;286:90–100.
20. Kumar PS, Abhinaya RV, Lashmi KG, Arthi V, Pavithra R, Sathyaselvabala V. Adsorption of methylene blue dye from aqueous solution by agricultural waste: Equilibrium, thermodynamics, kinetics, mechanism and process design. Colloid J. 2011;73:651–661.
21. Kumar PS, Fernando PSA, Ahmed RT, et al. Effect of temperature on the adsorption of methylene blue dye onto sulphuric acid-treated orange peel. Chem Eng Commun. 2014;201:1526–1547.
22. Kumar PS, Sivaranjanee R, Vinothini U, Raghavi M, Rajasekar K, Ramakrishnan K. Adsorption of dye onto raw and surface modified tamarind seeds: Isotherms, process design, kinetics and mechanism. Desalin Water Treat. 2014;52:2620–2633.
23. Gupta VK, Nayak A. Cadmium removal and recovery from aqueous solutions by novel adsorbents prepared from orange peel and Fe2O3 nanoparticles. Chem Eng J. 2012;80:81–90.
24. Nwadiogbu JO, Ajiwe VIE, Okoye PAC. Removal of crude oil from aqueous medium by sorption on hydrophobic corncobs: Equilibrium and kinetics studies. J Taibah Univ Sci. 2016;10:56–63.
25. El-Sayed G, Yehia MM, Asaad AA. Assessment of activated carbon prepared from corncob by chemical activation with phosphoric acid. Water Res Ind. 2014;7–8:66–75.
26. Azubuike CP, Okhamafe AO. Physicochemical, spectroscopic and thermal properties of microcrystalline cellulose derived from corncobs. Int J Recycl Org Waste Agr. 2012;1:9–11.
27. Hameed BH, Ahmad AA. Batch adsorption of methylene blue from aqueous solution by garlic peel, an agricultural waste biomass. J Hazard Mater. 2009;164:870–875.
28. Salazar-Rabage JJ, Leyva-Ramos R, Rivera-Utrilla J, Ocampo-Perez R, Cerino-Cordova FJ. Biosorption mechanism of methylene blue from aqueous solution onto white pine (Pinus durangensis) sawdust: Effect of operating conditions. Sustain Environ Res. 2017;7:32–40.
29. Yao YJ, Xu FF, Chen M, Xu ZX, Zhu ZW. Adsorption behavior of methylene blue on carbon nanotubes. Bioresour Technol. 2010;101:3040–3046.
30. Ali RM, Hamad HA, Hussein MM, Malash GF. Potential of using green adsorbent of heavy metal removal from aqueous solutions: Adsorption kinetics, isotherm, thermodynamic, mechanism and economic analysis. Ecol Eng. 2016;91:317–332.
31. Hameed BH, Ahmad AL, Latiff KNA. Adsorption of basic dye (methylene dye) onto activated carbon prepared from rattan sawdust. Dyes Pigm. 2007;75:143–146.
Fig. 2(a) Effect of contact time (Corncob = 10 g/L, Agitation = 150 rpm, Temperature = 30°C, pH = 6), (b) Effect of pH on the adsorption of MB (Corncob = 10 g/L, MB concentration = 0.005 g/L, Agitation = 150 rpm, Temperature = 30°C). 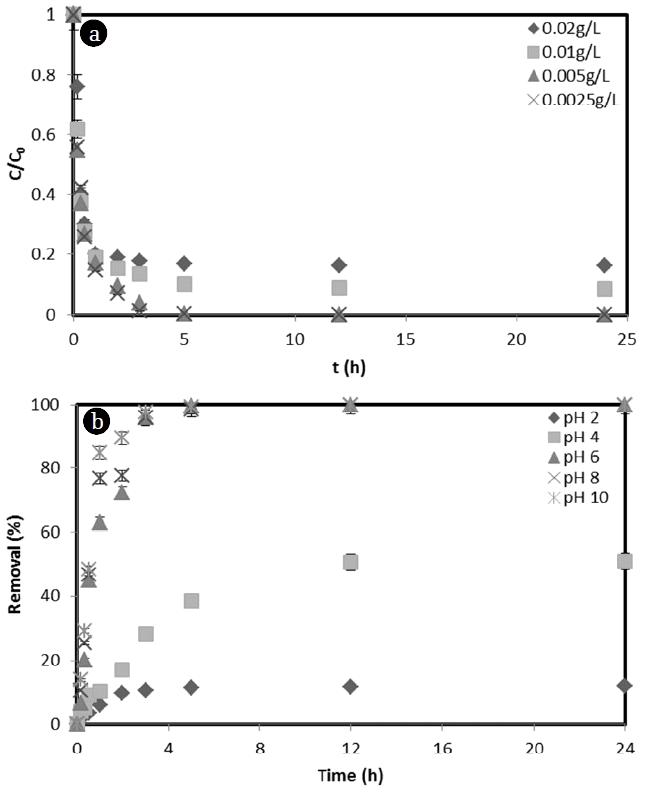
Fig. 3(a) Pseudo-first-order, (b) Pseudo-second-order kinetics, and (c) Intra-particle diffusion for the adsorption of MB. 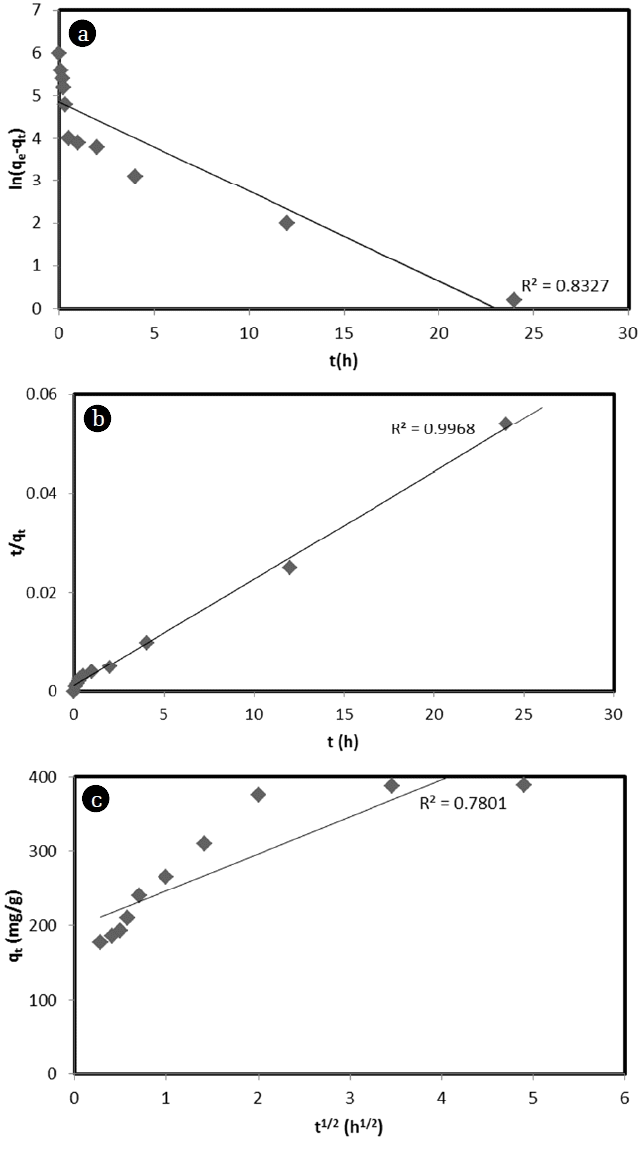
Fig. 4Linearized (a) Langmuir, (b) Freundlich, and (c) D-R adsorption isotherms for the adsorption of MB (Corncob = 10 g/L, MB concentration = 0.005 g/L, Agitation = 150 rpm, Temperature = 30°C, pH = 6). 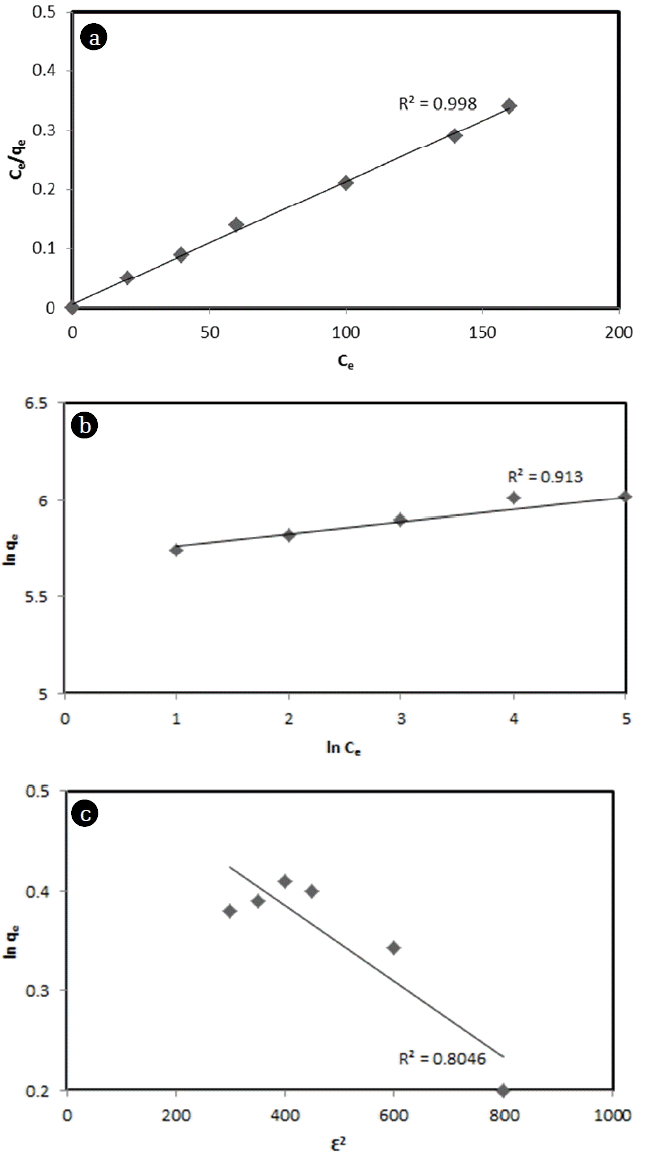
Fig. 5(a) Adsorption isotherm of MB at different temperatures, and (b) Application of van’t Hoff equation. 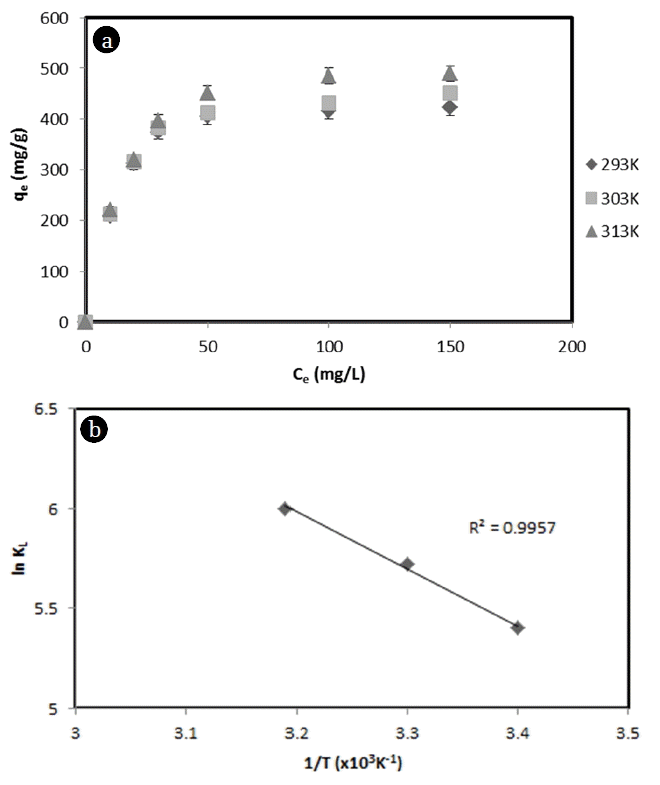
Table 1Elemental Analysis (wt, % of Dried Material) and Physical Properties of Corncob Table 2Kinetic and Isotherm Data for the Adsorption of MB by Corncob where qt and qe are the amount of adsorbed crude oil at time (t) and equilibrium, respectively. k1 and k2 are the pseudo-first-order and pseudo-second-order rate constant, respectively. Kd is the intra-particle diffusion rate constant (meq/g·h0.5) and I is the intercept. qe (mg/g) represents the sorption capacity at equilibrium, Ce is the equilibrium concentration in the solution, qm (mg/g) is the maximum adsorption capacity, and KL (L/mg) is the Langmuir adsorption equilibrium constant. KF is the constant that represents adsorption capacity related to bond strength, and 1/n is the constant indicating adsorption intensity. KF and n can determined from the linear plot of ln qe vs. ln Ce. KD (mol2/kJ2) is the D-R model constant related to the free energy of adsorption, Ɛ2 is polanyi potential (Ɛ2 = RT In (1+1/Ce), and E (kJ/mol) is the average adsorption energy. ΔG°, ΔH°, ΔS°, and T are Gibbs free energy, enthalpy, entropy, and absolute temperature in kelvin (K), respectively. Table 3Comparison of the Maximum Adsorption Capacity of MB Dye onto Agricultural Waste Adsorbents
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||