1. Introduction
Dyes are mostly used in many applications such as textile, printing, paper, food, and cosmetics as a colorant for the final products. However, dyes usage has been contributed to environmental pollution and health hazard. Nowadays, it is approximately about 700,000 metric tons of dyes are generated annually around the world [1, 2]. Since dyes wastewater has become the major effect on the environment, the used of new materials is a challenge in the membrane development process. The utilization of nanofiltration (NF) membrane has increased rapidly according to various applications although it is a relatively new type of the separation process. In wastewater treatment, membrane separation process was known to be among the most promising technology which offered an economical and environmentally approach. It has introduced a new perspective for the treatment of industrial effluents as well as industrial wastewater [3].
Having properties that combine size and electrical effects, NF membranes have solution-diffusion mechanism as present in the ultrafiltration, as those typically ruling transport in non-porous reverse osmosis membranes. NF membranes have pores typically near 1 nm in diameter and have fixed charges developed by dissociation groups have been used in the textile effluent treatment to lower the concentration of dyes. The results show that the NF membrane can promote the complete decolorization of effluent. According to the characteristics, NF membranes can retain multivalent complex ions and transfer small uncharged solutes and low charged ions [4, 5]. Moreover, steric hindrance and membrane solute interactions become the factors for the separation occurred in the NF membranes. Steric hindrance and non-electrostatic membrane-solute interactions were the main effects of the retention of uncharged molecules where their transport was taken over by convection due to a pressure difference and diffusion at different concentration gradient across the membranes [6].
In membrane separation process, most of the polymeric membranes have been prepared by phase inversion process. A homogeneous polymer solution is cast as a thin film or a hollow fiber shape and immersed into a coagulation bath. Between casting solution and non-solvent, the diffusional exchange of solvent and non-solvent can make the casting solution phase-separate to form a membrane with a symmetric or asymmetric structure. Addition of proper additive to the casting solution or the gelation media, introducing additional steps and coupling chemical reaction with phase separation process might give the right membrane properties. Surfactants, polymer, mineral fillers and non-solvents are examples of additives that have been used in casting solution during the preparation of polymer solution. The importance of these additives is to suppress and enhance the formation of macrovoids, enhance pore formation and improve pore interconnectivity and/or hydrophilicity [7].
Hence, the main goal of this study is to investigate the effect of polymer concentration and additive on permeate fluxes, dyes rejection, and morphology in polymeric phase inversion asymmetric NF membrane. The experiment has been conducted under the different experimental condition and the performance in term of morphology structures are characterized by using Scanning Electron Microscopy (SEM).
2. Materials and Methods
2.1. Chemical Materials
Polyethersulfone (PES) supplied by SOLVAY Advanced Polymer Company (RADEL A-300) was used as polymer materials. As a solvent, N-Methyl-2-Pyrrolidone (NMP) with analytical purity of 99.5% was purchased from Merck. The additive, Polyethylene Glycol (PEG 600) supplied by Merck. Ethanol and n-hexane both were purchased from Merck and used as the coagulation bath in membrane fabrication. Methyl Violet, Methyl Blue and Acid Orange 74 that has been used as synthetic dyes were purchased from Blulux Laboratory Reagents, Merck and Sigma-Aldrich.
2.2. Formulation of Polymer Solution
In general, a formulation of polymer solution contained of polymer, solvent, and non-solvent though sometime the addition of polymeric additive was preferred which can be known as multicomponent casting formulation. The multicomponent dope formulations are summarized in Table 1 below. In making polymer solution, PES was first dried for at least 1 d in a vacuum oven at a temperature of about 100 ± 2°C to leach out all water vapour. Firstly, the solvent and water were poured into the round bottom flask until the temperature increase to 50–60°C. When the temperature has reached 50°C, PES was added gradually until the entire polymer was dissolved before an additive was added. The solution was being stirred for 1 h and surfactant was added 3 h before the polymer solution was completely done.
2.3. Membrane Fabrication and Casting
In this study, asymmetric nanofiltration-surfactant (NFS) membrane was fabricated at room temperature (30 ± 2°C) with a variation of the polymer solution, ranging from 17 wt% to 21 wt%. The casting process was conducted as a small drop of the polymer solution was poured onto a glass plate. The glass plate was used as a support layer while casting knife was fixed at 150 μm at an appropriate casting rate (5–10 s). Subsequently, the fabricated membranes were immersed in a water bath for 24 h for coagulation process occurred before the membranes remained in ethanol for 1 d. Finally, the membranes were soaked in n-hexane for 2–3 h and kept at room temperature to remove residual organic compounds for 1 d before they are ready to be used.
2.4. Experimental Analysis
NF permeation test was conducted by using a simple permeation cell. Each membrane was subjected to be pressurized for compaction. The passages of the first 10 mL permeate was collected for analysis. Fig. 1 showed the schematic diagram of the experimental setup. The volume flux was calculated as below:
where Jv permeate flux (L/m2h), V is the volume of permeate solution collected (L), A is the effective membrane area (m2) and t is time (h). 400 kPa was chosen as operating pressure in order to study the effect of dyes rejection. Permeation flux test of dyes was carried out using different types of dyes (methyl violet, methyl blue, and acid orange) at three different concentrations (10 ppm,15 ppm and 20 ppm) before the rejection was calculated as follows:
where R is the rejection (%), Cp and Cf are the concentration permeate (mg/mL) and concentration feed (mg/mL), respectively. The experimental works were conducted with three times of analysis for triplication and all the experimental data of fluxes and rejection are the average values on the membranes performance.
2.5. Scanning Electron Microscopy (SEM)
Properties of the morphology for the fabricated membranes were analyzed by microscopic observation, scanning electron microscope (SEM- JEOL JSM-6360LA). SEM provides the visual information of the cross-sectional morphology and also the top surface of the membranes. Its images were taken under magnification ranging from 700X to 4000X. The membranes samples were dipped into liquid nitrogen and then coated with gold by using Auto Fine Coater (JFC-1600). The structural morphologies and membranes pores in terms of pore length, pore width were measured by an application supported by SEM.
3. Results and Discussion
3.1. Effects of Polymer Concentration on Performance
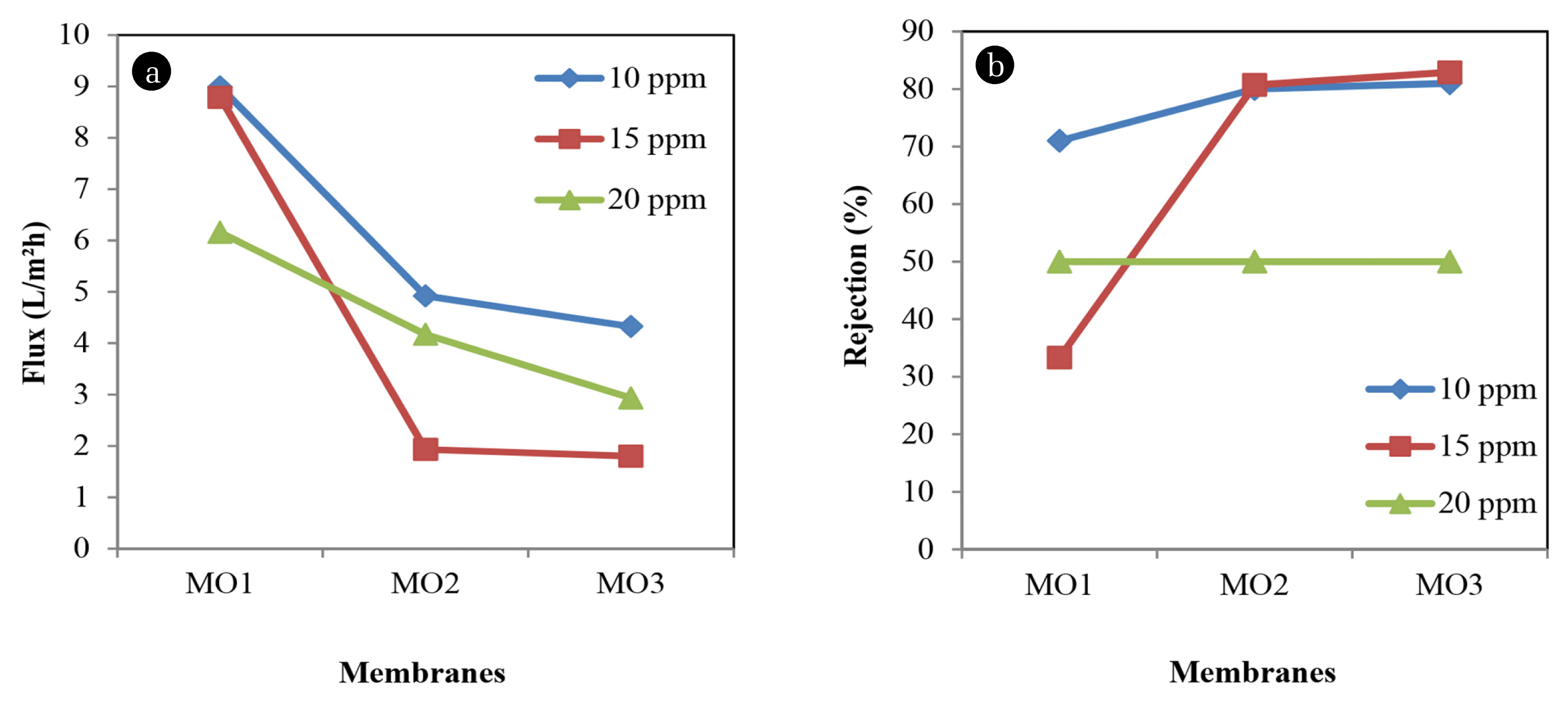
Fig. 2 shows the effect of polymer concentration on permeate flux and rejection of Methyl violet at three different dye concentrations. 10 ppm of dye solution shows the highest flux among other concentrations. However, the permeate flux keeps decreasing as polymer concentration increase from 8.9901 L/m2h to 4.3245 L/m2h. The rejection of 10 ppm of Methyl violet shows that as polymer concentration increase, the rejection will increase from 71% to 81%. At 8.7864 L/m2h, MO1 can reject only 33.3% of dye color for 15 ppm of Methyl violet. Since polymer concentration also plays an important role in this study, the color rejection of Methyl violet increases sharply to 82.9%. As dye concentration increase, the permeate flux keeps decreasing to 2.9318 L/m2h. However, at 20 ppm of Methyl violet, MO1 to MO3 resulted in the same rejection of about 50%. This finding provides evidence that in NF membrane process, concentration plays an important role to determine the level of performance of the prepared membranes. As can be seen in Fig. 2, permeate flux always higher at lower polymer concentration and decreasing gradually when polymer concentration increase, as well as Methyl violet concentration increases.
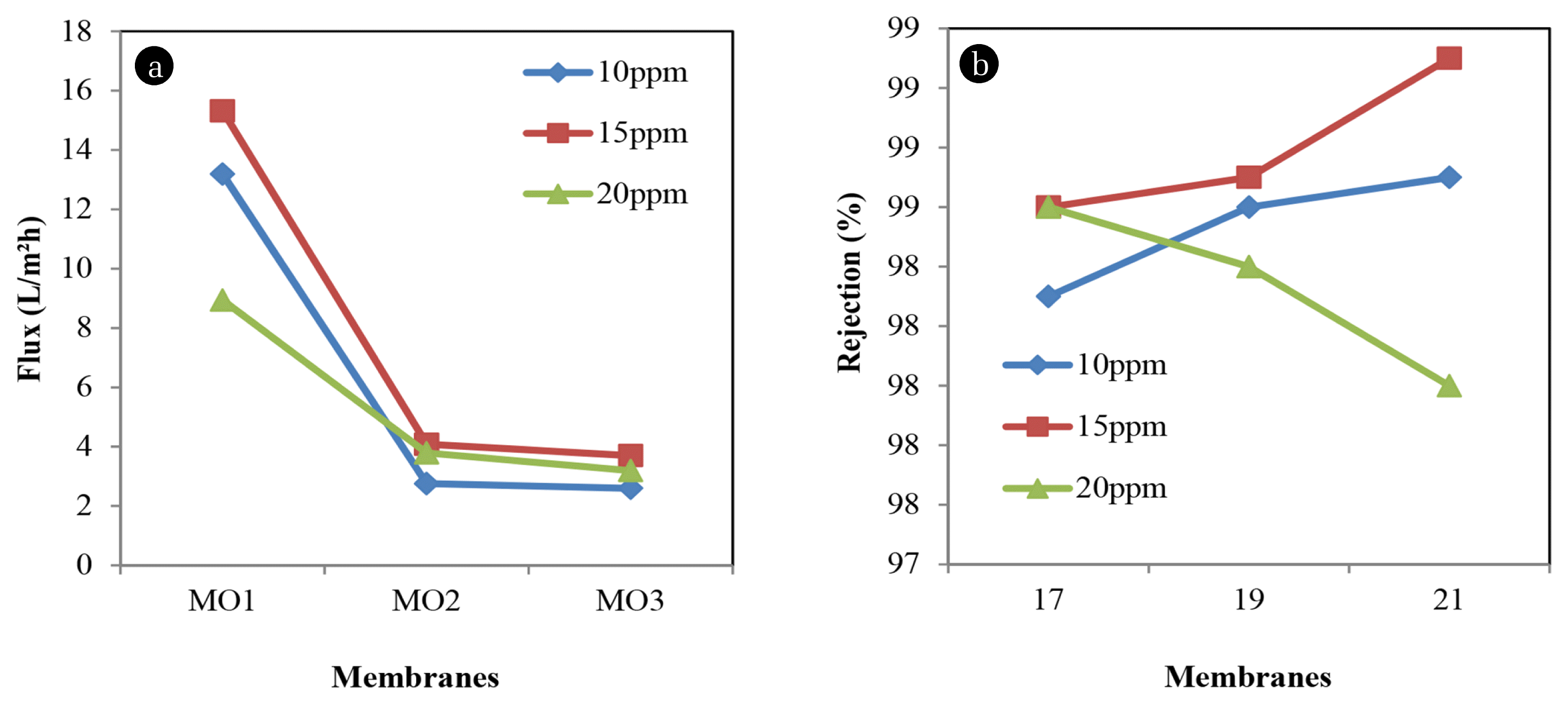
Fig. 3 shows experimental data of permeate flux and removal of Methyl blue using NF membrane at different polymer concentration. The highest permeate flux was obtained from the lowest polymer concentration at the lowest dye concentration. At 13.1886 L/m2h, MO1 can remove dye of about 98.3% at 10 ppm Methyl blue concentration. The results show that the permeate flux gradually decreases as polymer concentration increase until 2.5969 L/m2h for MO3 and increase the rejection up to 98.7% for the same dye concentration.
Research has been made to show that concentration of feed solution is the one of the parameters that affect the membrane performance [8]. At 15 ppm of dye concentration, the permeate flux shows the same results where the flux decrease when polymer concentration increase from 15.3193 L/m2h to 3.6984 L/m2h. Other than that, the rejection of 15 ppm dye concentration increasing up to 99.1% for MO3. This finding indicates that efficiency of NF membrane was proven to remove dye effectively. Membrane performance in terms of permeation flux and rejection of 20 ppm dye concentration keep decreasing. MO3 with highest polymer concentration in the formulation shows 3.1958 L/m2h of permeation flux and 98% rejection.
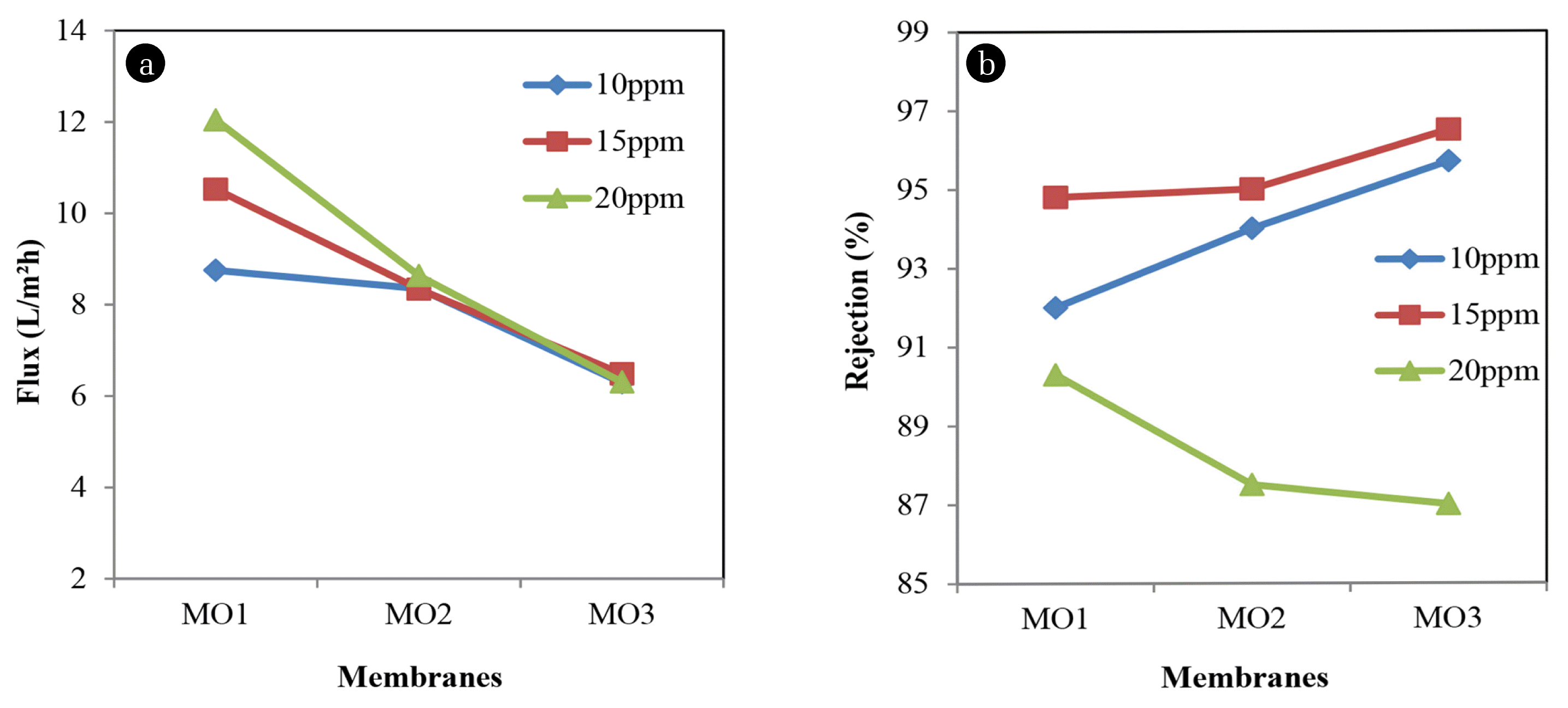
Fig. 4 presents the results of permeation flux and percentage rejection of Acid orange respectively. For 10 ppm dye concentration, at 8.7539 L/m2h, MO1 gives rejection of about 92%. As polymer concentration increase, the permeation fluxes keep decreasing to 6.2924 L/m2h and rejection increasing up to 95.7%. The same trending happens with 15 ppm of dye concentration where the highest polymer concentration, MO3 gives the lowest permeation flux of about 6.4873 L/m2h and gives rejection of 96.5%. Research has been found that concentration of feed solution also played an important role in determining the performance of membrane using dyes solution.
At 20 ppm of dye concentration, the permeation fluxes keep decreasing as polymer concentration increase from 12.0452 L/m2h to 6.3077 L/m2h. However, the rejection is decreasing as polymer concentration increase. At high polymer concentration, MO3 gives only 87% of dye rejection. This indicates that the high concentration of Acid orange will decrease the percentage. Other than that, morphological structure of membrane also affects the membrane performance in terms of numbers of pores, finger-like substructure and support layer that helps the movements of molecule throughout the membrane.
3.2. Effects of Addition of Additive on Performance
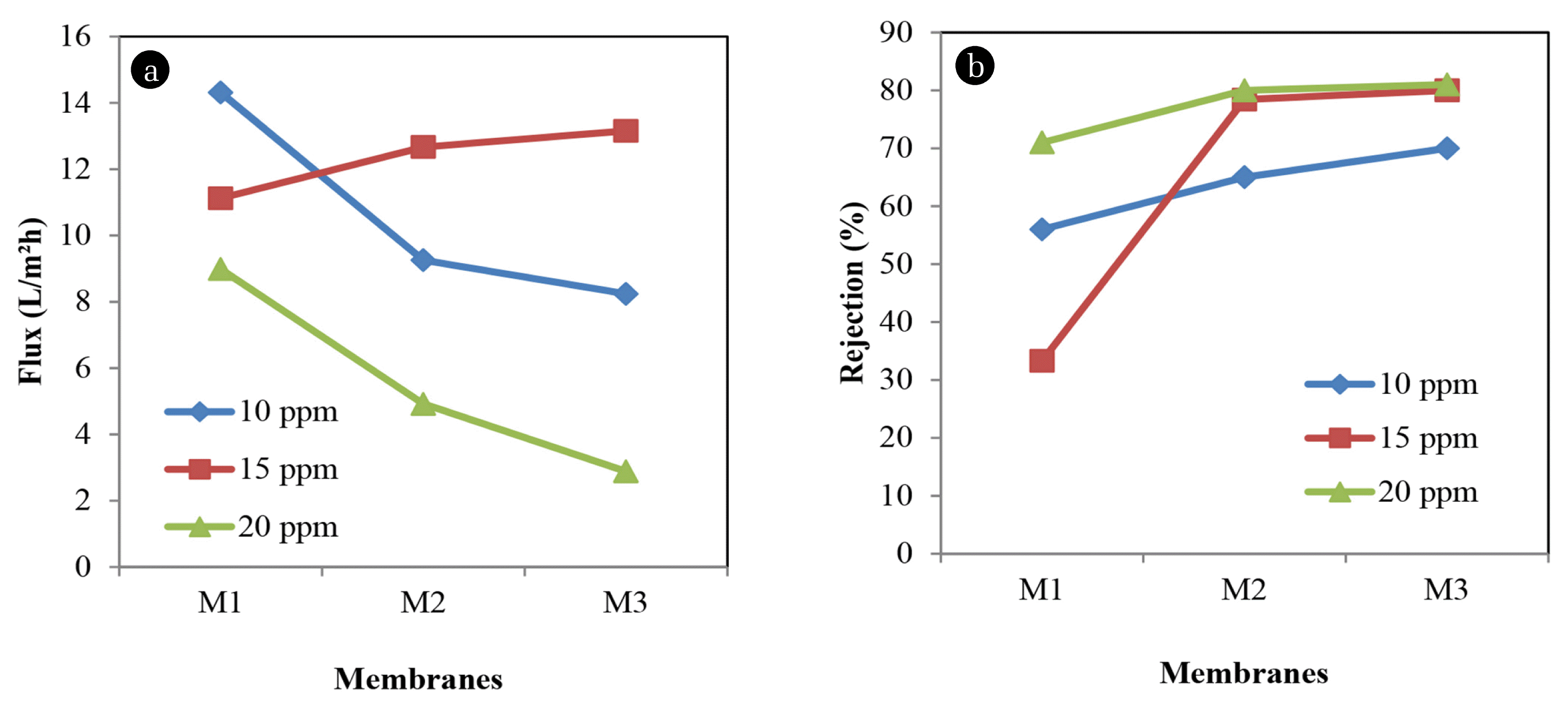
For further study on the performance of NF membrane with the addition of additives, Methyl violet was used prior to test as shown in Fig. 5(a) and (b). Since 10 ppm Methyl violet is the least dye concentration, it always gives the higher permeation flux among others concentration of about 14.3149 L/m2h at the lower polymer concentration. As polymer concentration increase, the permeation flux of 10 ppm Methyl violet decrease to 8.2376 L/m2h and rejection gradually increase to 70%.
The result of Methyl violet testing would be different for 15 ppm dye concentration where the permeation flux increases as well as rejection. This is due to the changes in membrane structures that lead to the high permeation flux and high rejection of Methyl violet. M1 was observed to have 11.1223 L/m2h of permeation flux and have a low rejection of about 33.3%. The rejection of this dye concentration increases sharply up to 80% when polymer concentration increases. When polymer concentration increase, the permeation flux of 20 ppm Methyl violet decrease from 8.9901 L/m2h to 2.8852 L/m2h and rejection was increase from 71% to 81%.
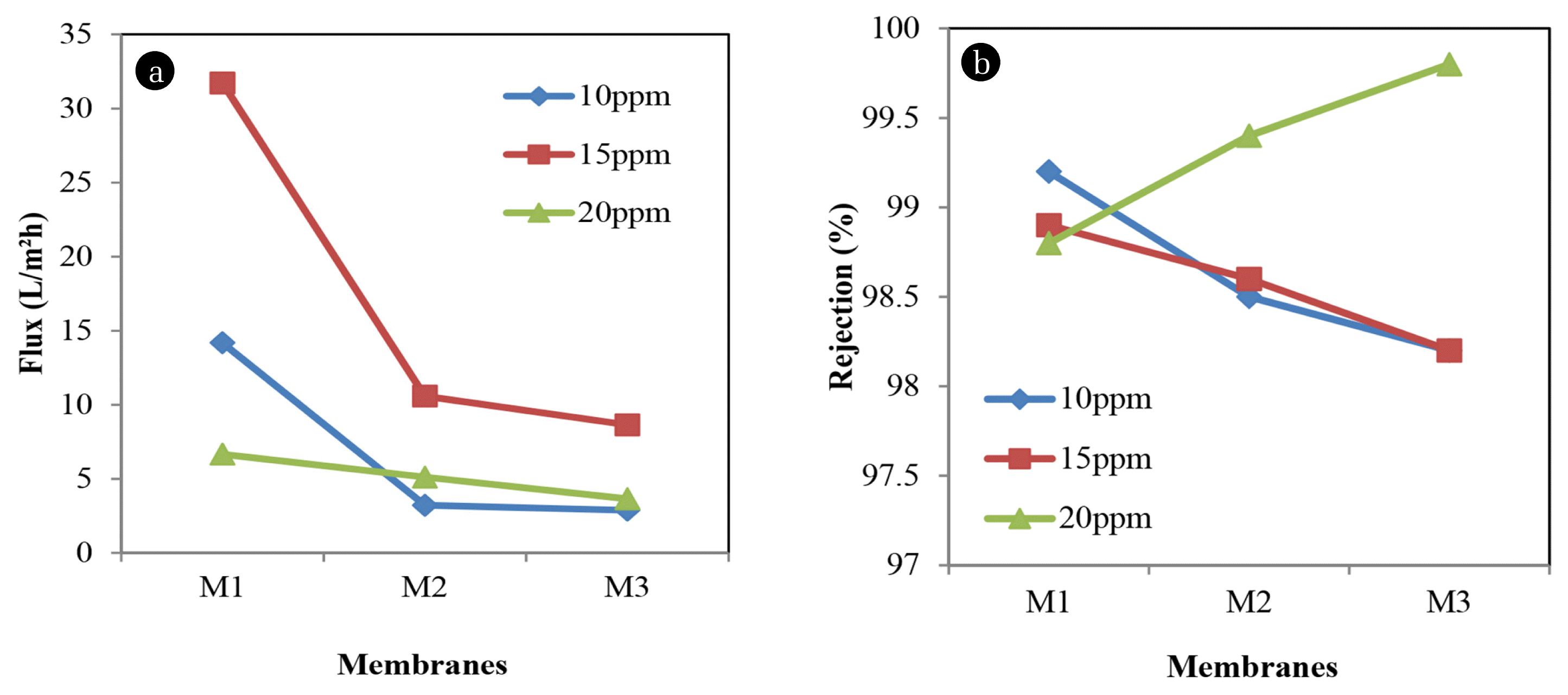
Fig. 6(a) and (b) presents the experimental data that have been done in terms of permeation flux and percentage rejection of Methyl blue. With 799.81 g/mol molecular weight, Methyl blue seems can reject almost 100% of dye content. At 14.1820 L/m2h, 10 ppm of Methyl blue rejects almost 99.2% of dye contents. This shows that at low dye concentration and low polymer concentration, membrane surface of M1 is thinner so that it can reject a high percentage of dyes.
As dye concentration increase, permeation flux of M1 drop to 6.6619 L/m2h. However, there is no significance decreasing in rejection where 20 ppm rejects almost 98.8%. Moreover, experimental also revealed that, as polymer concentration increase, the permeation flux would decrease. From M1 to M3, the permeation flux of 15 ppm dye concentration decrease from 31.7234 L/m2h to 8.6432 L/m2h. Rejection of 15 ppm dye concentration also shows a decrease about 0.7% from 98.9% to 98.2%. This shows that, when polymer concentration increases, the percentage rejection is not affected well. With the addition of the additive, M2 shows increasing of permeation flux for 20 ppm dye concentration from 3.791 L/m2h to 5.114 L/m2h. This revealed that the efficiency of pore forming additives in changing the membrane properties.
Furthermore, experimental data in Fig. 6 shows that, at 20 ppm dye concentration, the permeate flux was decreased to 3.6501 L/m2h. A researcher has discovered in his study that concentration plays important aspects in NF membrane [8]. Yet, at 20 ppm dye concentration, rejection of Methyl blue keep increasing up to 99.8% which indicate the complete separation of dye compounds from dye solutions.
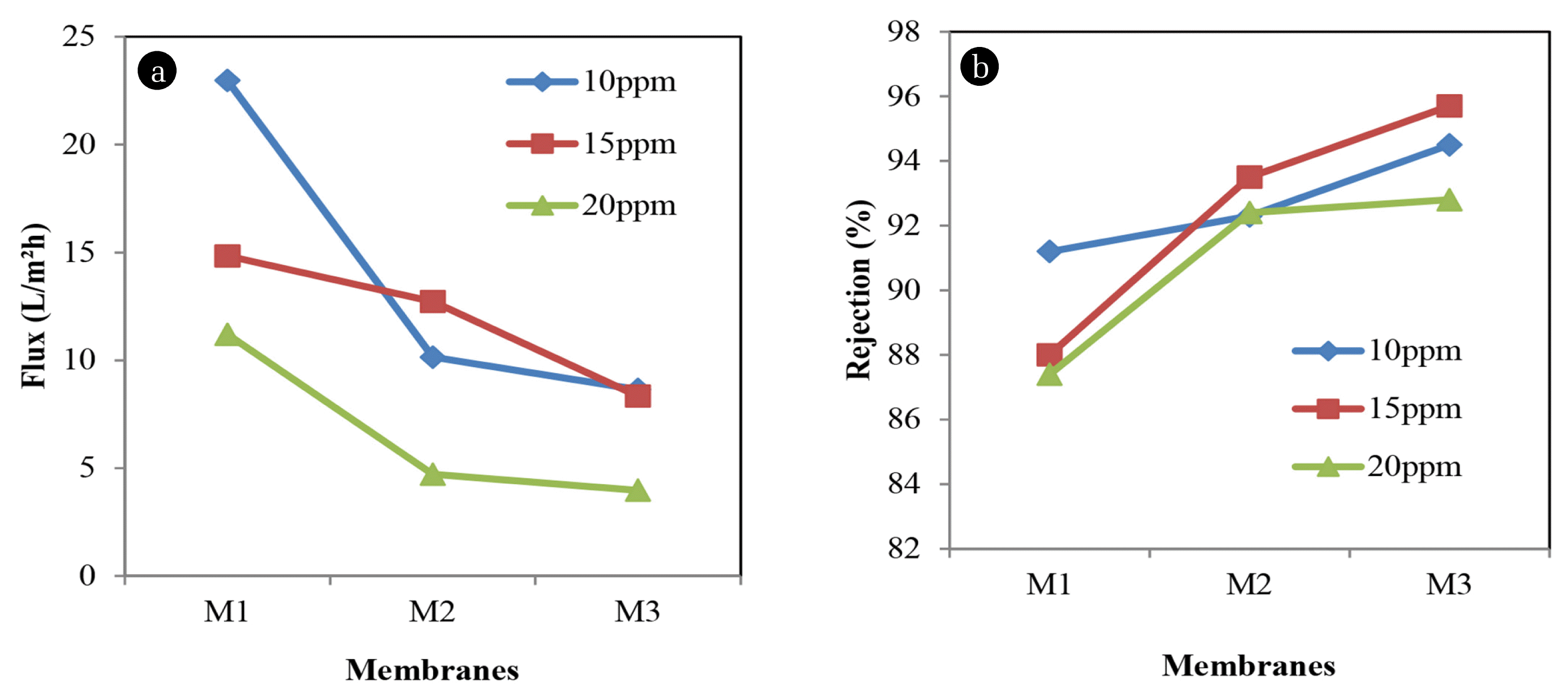
Fig. 7(a) and (b) illustrate the permeate flux and percentage rejection of acid orange respectively. At 22.9721 L/m2h, M1 at 10 ppm dye concentration gives rejection of about 91.2%. As polymer concentration increase, the rejection increase up to 94.5% while permeate flux decreased up to 8.6461 L/m2h. Moreover, as dye concentration increase, Acid orange shows a decrease of permeate flux as well as rejection. M3 shows permeate flux of about 3.9776 L/m2h and rejection of about 92.8%. Besides, this phenomenon might due to the concentration polarization and osmotic pressure that leads to fouling on the membrane surface and cause the decreasing of permeate flux and increasing in dye rejection [9]. Thus, the study has proven that using a high concentration of Acid orange resulted in decreasing of permeate flux and increase the rejection up to 95.7%.
3.3. Morphological Study
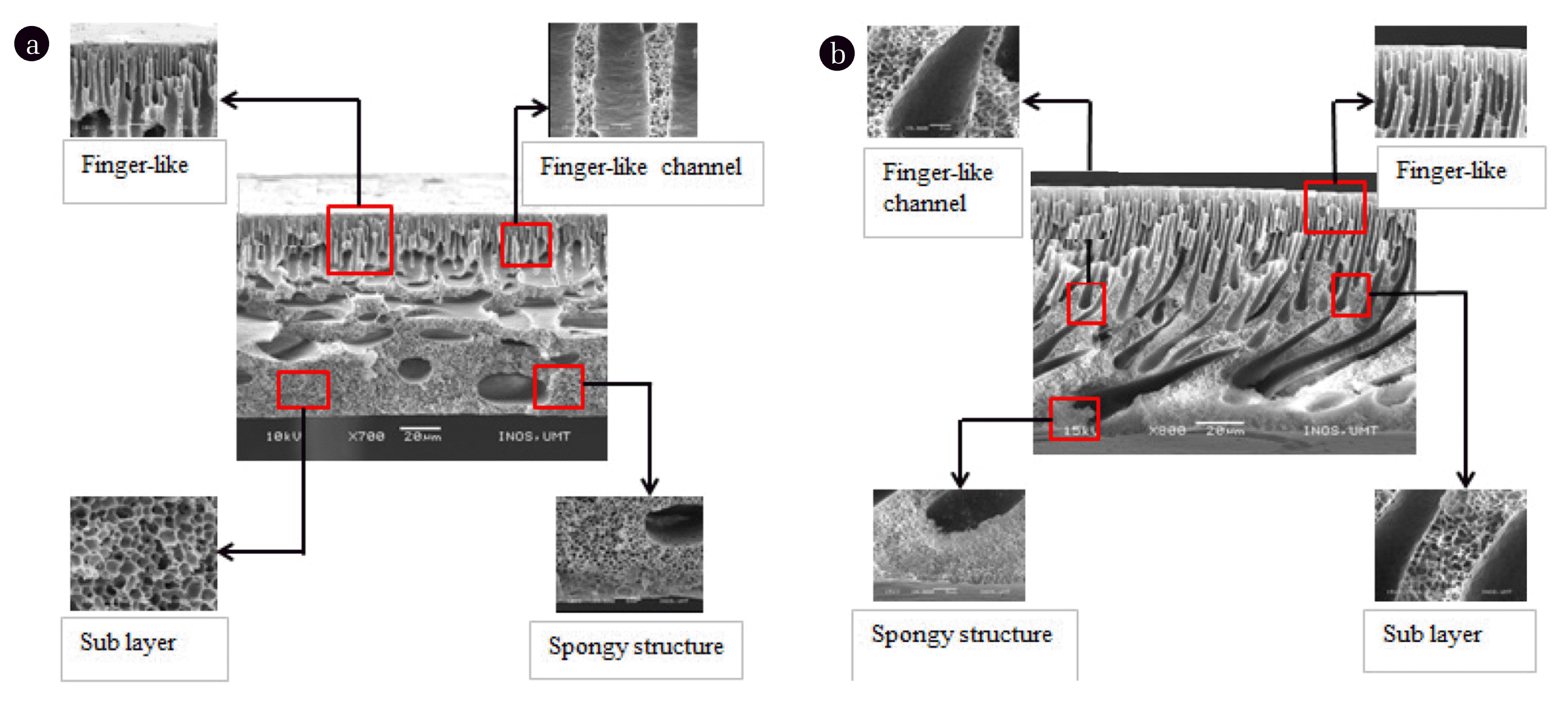
As shown in Fig. 8(a), it can be seen that an asymmetric NF membrane consists of dense top layer, finger-like structure, and membrane sub-structure. Moreover, macrovoids also has been increased. The same form of membrane support layer existed where PES form thick membrane support layer which makes the membrane more stable. Fig. 8(b) shows denser top layer and thick membrane surface are produced. It was reported that addition of additive could be one of the major factors influencing the membrane performance [10]. The additive is used in order to have an optimal membrane structures that leads to membrane performance-properties. High polymer concentration has shown the least macrovoids formation.
Other researchers found the similar structure in their study that, the addition of PEG 600 as the additive will form a finger-like structure that almost penetrates throughout the membranes and improve the membrane pore interconnectivity [11]. At the 21 wt% of polymer concentration, it shows good membrane structure with spongy support layer that eases the separation process throughout the membrane.
4. Conclusions
From this study, it was found that PES has been confirmed as the promising polymeric material as it showed asymmetric structure via phase inversion technique. Meanwhile, PEG 600 has been known as a better pore former than other additives. It has increased the rejection of dyes up to 99.8% and influenced the formation of the denser top layer and thick membrane surface as has been seen in a morphological study. Thus, this study significantly contributes to new knowledge and also in treatment wastewater mainly for dyes industry.