1. Introduction
The ever-growing water demand has not been satisfied domestically and globally by water supply at the presence of abnormal climate changes such as uneven distribution of droughts and floods. Water is no longer a ‘gift’ from nature but is considered as a ‘life-support resource’. As water is regarded as a renewable resource, compared to non-renewable resources such as coal and oil, various studies have been carried out to secure water resources over the recent decades [1–3]. New water resources can be created through three key words: water reuse, new technology development and water strategy education which reflect the importance of cyclic processes. Reused wastewater could be regarded as a new water resource, especially when it is used in regions where freshwater is scarce [4]. Especially, coal seam gas (CSG) water, which needs to be pumped out from coal seams for CSG production, had not been considered as a usable water resource in Australia until a new policy on CSG water management from Queensland State Government was recently released to encourage the beneficial used of CSG water [5]. CSG water, to be discharged, has been usually treated in reverse osmosis (RO) plants which require extensive and expensive pre-treatment. However, current low gas prices have been a great driver for relevant industries to seek for alternative cost-effective technologies in the aspect of its beneficial use and fit-for-purpose usable water production.
To design processes for usable water production, the sections of functional materials and application methods need to be viable to remove salts and total dissolved solid (TDS), hardness (mostly caused by the presence of Ca2+ and Mg2+), carbonates (i.e. NaHCO3), colour, petroleum compounds and organic matter (as total organic carbon (TOC)) [3]. In this study, a coagulation reactor, a pore control fiber (PCF) filtration system (which need relatively small footprints [6]) and a RO system (which is effective in removing ionic compounds [7, 8]) were combined to treat CSG water. Linking with our previous results [9] that analysed Australian government policy on the beneficial usage of CSG water [10], this paper presents a CSG water treatment option which is based on domestic technologies. A coagulation reactor was placed to further remove SS in front of the PCF. As Al ion in poly aluminium chloride (PAC) has recently been revealed to be one of substances that induce dementia, a Fe(III) type of Polytetsu as an alternative, which was developed by a domestic company, was selected and tested in this paper. Based on the test results, the potential of the combined system was evaluated for a usable water production system from CSG water. Also, this paper presents the treatment efficiencies in terms of water quality requirements for water supply purposes such as industrial, agricultural and domestic uses.
2. Materials and Methods
2.1. Pilot Plant Construction and Operation
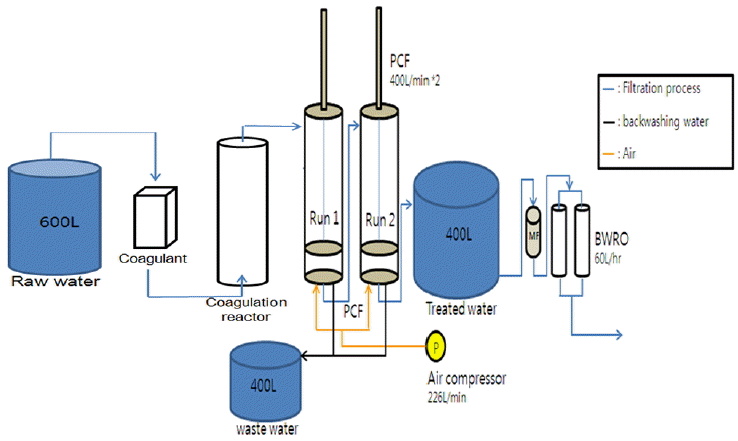
As shown in Fig. 1, a small-scale pilot plant was designed and constructed to treat CSG water, consisting of a coagulation reactor, a two-stage PCF system and a RO system, having a timer function and a backwashing option. The PCF was selected as it requires less capital cost and footprint than membrane separation units. And asymmetric porous polyethylene membrane was used for the RO system. The design treatment capacities of the PCF and RO systems were 400 and 51 L/h, respectively. The volume ratio of effluent over influent was adjusted to be 12.75%. The operating conditions are listed in Table 1. According to the average water quality in CSG water management policy [5] as listed in Table 2, a simulated CSG water was made and used in this paper, having water qualities listed in Table 3.
2.2. Coagulants
Due to its simplicity and effectiveness, coagulation-flocculation processes are regarded as one of the most important and widely used treatment process of raw water [11] and wastewater [12]. To improve the SS removal performance of the PCF system [11–13], along with a common type (Al(III) type of PAC), a newly developed Fe(III) type of Polytetsu by Cheong Woo ACE Co. [14] in South Korea was selected and their properties are listed in Tale 4. As Al ion in PAC is one of substances that induce dementia, a lot of effort has been devoted to seek for alternative coagulants. As part of our endeavours, a Fe(III) type of Polytetsu was selected to find out its applicability. Pre-determined conditions for the coagulation-flocculation process were used in this study. After confirming the coagulation areas, jar tests were carried out to obtain the coagulation efficiency with the concentration ranges of 10, 20, 30, 40 and 50 ppm at pH of 5, 7 and 9 for PAC and 4, 7 and 10 for Polytetsu [15].
3. Results and Discussion
3.1. Analysis of CSG Water
According to the average CSG water qualities in Table 2 [5, 16], the sum of sodium, calcium, magnesium, potassium, boron, chloride and TOC concentrations is about 4,688 mg/L. As the TDS concentration is 4,450 mg/L, the difference (238 mg/L) from the sum is a portion that was not detected as TDS. If TOC (248 mg/L) is excluded, the sum is similar to the TDS concentration. This suggests that the TOC in Table 2 may not be ionic organic carbons. This factor was used for the system design.
3.2. Prediction of Coagulant Molecular Weights
The molecular formula of the selected PAC is [Al2(OH)nCl6-n]m (1 ≤ n ≤ 5, m ≤ 10, alkalinity: n/6×100) with 17% of Al2O3. As the selected PAC has more than 35% of alkalinity, n is expected to be more than 2. However, considering the fact that the pH region in the coagulation gets narrower with higher alkalinity, n would be 2 or 3. As the PAC is an inorganic coagulant, the degree (m) of polymerization to increase the molecular weight would be maximum 10. Therefore the molecular weight distribution can be estimated between 2,990 and 3,150 (average 3,000). Also, [Fe2(OH)n(SO4)3-n]m (0 ≤ n ≤ 2, m = f(n)) as a Polytetsu was used. As it has 3D configuration with some of SO4−2 being substituted with OH− and the alkalinity is high, n is estimated to be 2 with m being 10 (the same as PAC), resulting in the molecular weight of 2,416 [17, 18].
3.3. Linear Regression between Concentration and Turbidity of Coagulants
As the relationship between feed concentration and turbidity of the selected coagulants is an important aspect of coagulant characteristics [19, 20], it was obtained with respect to pH and the linear regression results are listed in Table 5. The PAC was found to have slopes of 68.84, 70.47 and 67.00 with R2 of 0.940, 0.918 and 0.930 at pH of 4, 7 and 9, respectively, indicating that the impact of pH is insignificant. This could be due to the fact that pH affects the concentrations of ionic species in aqueous solution. Similarly, a consistent trend was observed for the Polytetsu having slopes of 108.84, 117.82 and 107.15 with R2 of 0.958, 0.950 and 0.970 at pH of 5, 7 and 10, respectively. Also, the turbidity of Polytetsu was more rapidly increased as the feed concentration rises, compared to that of PAC.
3.4. Coagulation Areas of PAC
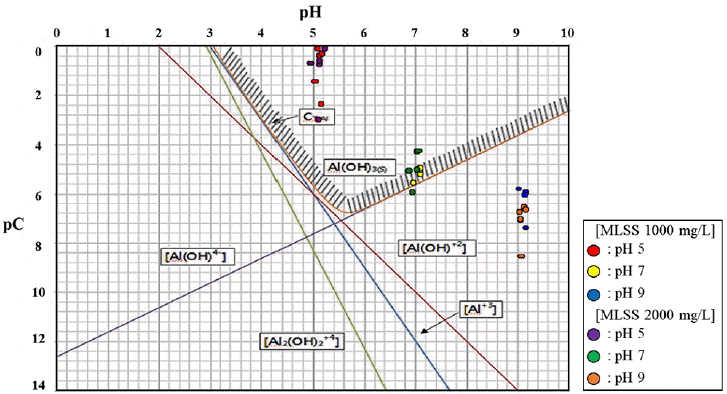
Possible types of Al3+ complexes in an aqueous solution were considered as below and their concentrations are plotted with respect to pH in Fig. 2.
As can be seen in Fig. 2, all coagulant concentrations are inside the coagulation area at pH between 5 and 7 whereas they are located at the outer area at pH 9. In the case of high alkalinity, the high concentration of OH− would yield aqueous dispersion of ionic species, leaving the coagulants out of the coagulation area. The coagulation efficiency was found to be optimum at pH 7. Therefore, if PAC is used for the pilot plant, then pH needs to be close to 7 [21]. This is one of design criteria.
3.5. Coagulation areas of Polytetsu
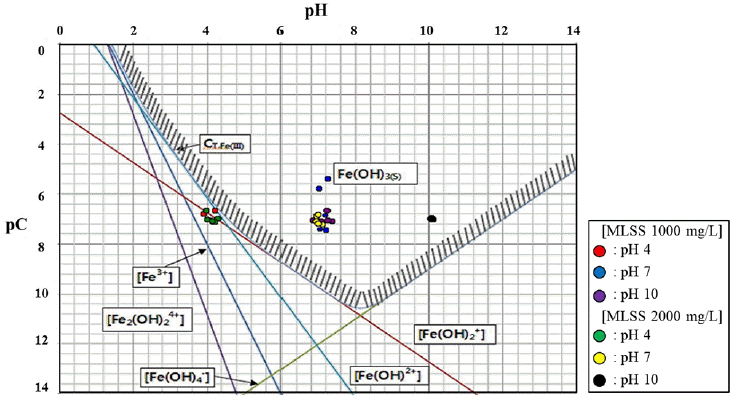
Similar to the case of PAC, possible types of Fe3+ complexes in raw water with SS of 1,000 and 2,000 ppm were selected as below and their coagulation area is platted at broader pH (4, 7 and 10) than for PAC in Fig. 3.
The Polytetsu seems to be out of the coagulation area at pH 4. However, as Fe(OH)2+, Fe(OH)2+ and Fe(OH)3(S) are present at similar concentrations, a similar coagulation efficiency was observed, regardless of the pH range used in this study [22]. This indicates that coagulation still takes place even if a certain amount of ionic complexes is present. Therefore, unlike the PAC, it is confirmed that the Polytetsu is always in the coagulation area at the pH range studied. Thus, although the Polytetsu with Fe(III) ion has slightly lower estimated molecular weight than the PAC, the former can be applied at broader pH range than the latter. The supernatant after coagulation was about 10−7 M, indicating that the Polytetsu is better than the PAC in terms of optimum coagulation concentrations. Therefore, it can be said that a coagulation system with a Fe(III) type of Polytetsu in front of the PCF system is an effective option [6] for the operation of the pilot plant constructed in this study.
3.6. Preliminary Operation of Pilot Plant
The PCF/RO system was operated using a Fe(III) type of Polytetsu coagulants and the preliminary results are listed in Table 6. As the Polytetsu coagulant in front of the PCF/RO system made the floc size enlarged, more than 99% of SS were removed through the PCF process. After 20 min of operation, the SS removal efficiency was stabilized, suggesting that 20 min would be an optimum condition, considering the energy consumption. However, the removal efficiencies of organic, total nitrogen (T-N) and total phosphorous (T-P) were found to be low (30.4–32.0, 13.0–25.0 and 6.0–20.4%, respectively) with the PCF process while most of them were removed to undetectable levels with the RO system. In particular, along with a decrease in conductivity, as the TDS derived from salts was removed in the RO system, it is necessary to have a RO system to meet the TDS discharge level (< 500 mg/L) for drinking water [5]. In addition, extensive pre-treatment processes such as the PCF in this study are required to reduce RO membrane fouling. As listed in Table 7, OH−, HCO3− and CO32− were measured for M and P alkalinities [23–25]. Having OH− undetected, HCO3− was found to be dominant, being removed up to 97–98 % with the RO process. Especially, there was no noticeable change in ORP, which can be used to predict oxidation-reduction reaction at the early stage operation. This suggests that an additional process is required to oxidize non-ionic organic carbons (detected as TOC). Therefore, for the operational demonstration of the pilot plant using these results, the effects of carbonate on organic matter oxidation, CSG water storage and transport will be investigated in near future.
4. Conclusions
In this paper, a combined system with a coagulation reactor, a two-stage PCF filtration system and a RO system was constructed and tested for CSG water treatment. To improve the PCF filtration efficiency, a Fe(III) type of Polytetsu was found to be a better option than a Al(III) type of PAC due to its greater coagulation efficiency and applicability at a broader range of pH than the latter. The PCF filtration process removed 99% of SS while the RO system removed most of organic, T-N and T-P. In particular, along with a decrease in conductivity, the TDS derived from salts was removed in the RO process. To meet the TDS discharge standard (< 500 mg/L) for drinking water, it is found that a RO process is still required. As the preliminary operation results showed no noticeable change in ORP, an additional process is required to oxidize non-ionic organic carbons (detected as TOC). From the measurement of OH−, HCO3− and CO32− in terms of M and P alkalinities, HCO3− was a dominant compound, being removed 97–98% with the RO process. OH− was not detected. Thus, for the operational demonstration of the pilot plant using these results, the effects of carbonate on organic matter oxidation, CSG water storage and transportation will be investigated in near future. Also, a range of fit-for-purpose system designs for beneficial uses of CSG water will be presented in the next study.