1. Introduction
Ammonia (NH3) is a valuable chemical resource for use in industry as a fertilizer, refrigerant, and to refine petroleum [1]. However, NH3 is also a toxic gas that has a pungent odor and it has potentially harmful effects on the public [2]. When the NH3 content in the atmosphere exceeds 35 ppm, it impacts the health of humans and animals [3]. In addition, an aerodynamic study of particulate matter (PM) in the presence of NH3 found that it played a vital role in the formation of the fine fraction of the aerosol (PM2.5) [4]. Hence, NH3 adsorption is of great importance for human health.
Ideal adsorbents with high surface areas and small pore sizes are preferred for high capacities to remove contaminants. Research into such porous materials has gradually addressed these applications with respect to NH3 adsorption or filtration [5–7]. The traditional inorganic adsorbents, including activated carbon, alumina, silica gel, and 13X zeolite, have low adsorption capacities in the range from 2.3 to 12.0 mol/kg (37 to 192 mg/g) [7]. In recent years, the number of available nanoporous materials, such as mesoporous silica (MS) has increased substantially and emerged as a gas adsorbent alternative to zeolite and activated carbon. The highly regular pore structure and internal surface area of ordered MS make these particles attractive substrates that can be tailored for NH3 adsorption [8]. For example, MS particles showed very high uptake capacities and selectivities for CH4 [9], CO2 [10], H2S [11], NH3 [12], and volatile organic vapors [13]. Few studies on the removal of hazardous gases by adsorption using MA are reported, even though MA and MS share similar pore properties and high thermal stabilities, as seen in comparison to reports about MS in the literature [14, 15].
Therefore, herein, MA prepared by a templating method was used as an NH3 adsorbent. In previous studies [16, 17], MA showed a high adsorption capacity for metal ions (Hg2+, Pb2+, and As3+) from the aqueous phase. We found that the surface area provided by different adsorbents did not significantly affect the adsorption capacity, and in fact, the key factor was a uniform pore size and an interlinked pore system. Since those particle properties of MA enhanced the adsorption capacity of the substrate for the removal of metal ions, MA might also be helpful to use as gas-adsorbent. Because zeolite, activated carbon and silica nanoparticles were used as commercial gas adsorbents in real-world applications, we also evaluated these materials for their NH3-adsorption capacities.
2. Materials and Methods
2.1. Materials
As described in our previous reports [16–18], stearic acid and aluminum sec-butoxide were used as the anionic surfactant, and an aluminum precursor was used to prepare MA. The aluminum precursor and stearic acid were dissolved separately in sec-butyl alcohol, and then the two solutions were mixed. A small amount of water was slowly added dropwise to the mixture, generating a white precipitate. After further stirring for 24 h, the resulting materials were calcined for 3 h at 500°C. The molar ratio of this reaction mixture was 1 Al(sec-BuO)3: 0.2 C17H35COOH: 0.04 NaOH: 5 sec-BuOH: 4 H2O. Zeolite (Ze, < 45 μm, CAS#1318-02-1), activated carbon (AC, 100 mesh), and silica (Si, 12 nm) were purchased from Sigma-Aldrich (St. Louis, MO, USA) and used without further treatment as commercial adsorbents. The porosities of the adsorbents were analyzed by transmission electron microscopy (TEM, JEM-2010, Jeol, Japan). N2 adsorption/desorption experiments were carried out using a BELSORP-mini volumetric adsorption measurement instrument (BEL, Japan), and pore size distributions were calculated using the Barret–Joyner–Halenda (BJH) pore size and volume analytical model on the desorption branch.
2.2. Adsorption of NH3
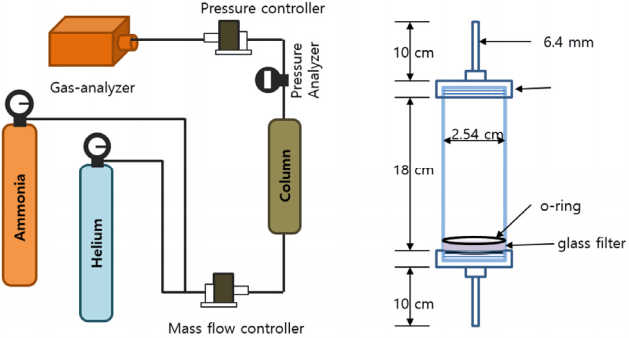
Helium (99.999%) and NH3 (99.999%) were used as carrier and target gas molecules, respectively. As shown in Fig. 1, the experimental setup for measuring the isotherm consisted of a stainless-steel column (18 cm length and 2.54 cm inner diameter) with 5 g of packed adsorbent sample, a mass flow controller (max. 5 L/min), a pressure controller (max. 5 bar), and a continuous gas analyzer (AR8500, Smart Sensor, USA). The gas hourly space velocity was fixed as 28 min−1. The desorption of NH3 adsorbed on MA was carried out at 400°C at 5 L/min He for 2 h. After regeneration, three recycling tests were carried out at a given pressure. In addition, to define the Gibbs free energy for the adsorption of NH3 on MA, the temperature of the reactor was changed from 25 to 5°C.
3. Results and Discussion
3.1. Characterizations of Adsorbents
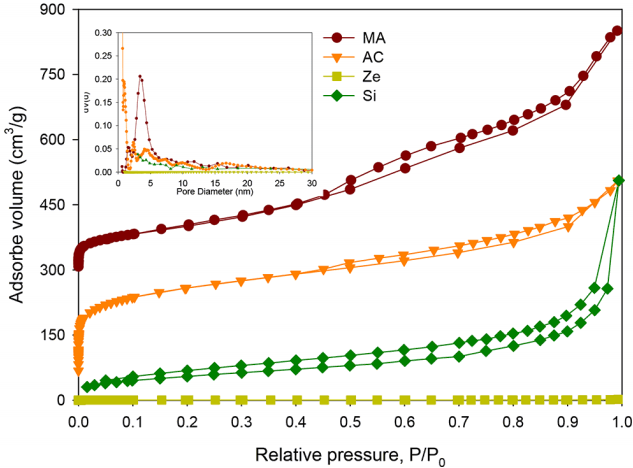
As shown in Fig. 2 and Fig. 3, MA showed a pore diameter of 3.4 nm with a narrow and uniform pore size distribution (DFWHM ~ 2 nm). In the N2 adsorption/desorption isotherm of MA, a typical type of IV isotherm of mesoporous porosity and a hysteresis loop in the range 0.4–0.8 P/Po was observed, showing both the framework and textural porosity of the substrate. The framework porosity was in the range 0.4–0.7 P/Po, indicating that the porosity was contained within the uniform channels of the templated framework, whereas the textural porosity in the range 0.8–1 P/Po was indicative of the porosity arising from the non-crystalline intra-aggregate voids and spaces formed by interparticle contacts [16]. The TEM images (Fig. 3(a)) of MA showed a sponge-like porous appearance and an interlinked pore system, consistent with the analysis for the N2 adsorption/desorption result of MA. An interconnected pore structure has an advantage of a reduced diffusion limitation and an enhanced molecular accessibility to the inner surface.
However, other adsorbents (AC, Ze, and Si samples) showed irregular and broad pore size distributions, as shown in Fig. 2. The AC sample had a small pore at the inner surface and a large surface area (1,024.2 m2/g), but both the non-uniform pore structure and very broad pore distribution in the range 1–25 nm did not match with the physical properties of ideal adsorbents. A molecular sieve such as the Ze sample generally has a very small pore (2–10 Å), which is too small to be able to adsorb NH3 into the inner surface of Ze. The Si sample consisted of single particle with a size of 12 nm and aggregates of these particles are depicted in Fig. 2(d). Because the void fraction originated from aggregation between the primary particles, it acted as pores in the N2 adsorption/desorption test. The pore properties of all the adsorbents are listed in Table 1. Therefore, NH3 might be adsorbed on the outer surface of Ze and Si samples, but on both inner and outer surface of MA and AC samples.
3.2. Adsorption Equilibrium Study
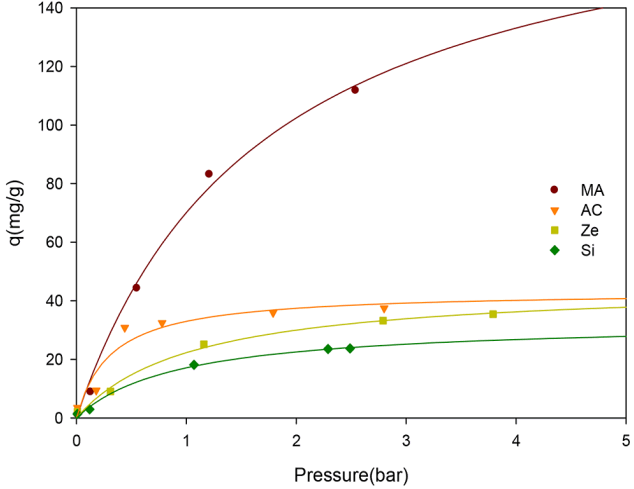
Applied pressure is the controlling parameter to adjust the gas uptake on adsorbents. When increasing the applied pressure of the NH3 gas, the uptake capacities of adsorbents generally increased to saturation levels at high pressure. As shown in Fig. 4, the saturation curve for all adsorbents followed a typical Langmuir isotherm. While the uptake of NH3 gas on MA increased steadily up to 3 bar, AC, Ze, and Si samples showed rapid saturation after 1.5 bar. NH3 molecules were first adsorbed on the outer surface of the adsorbent at a relatively low pressure and then gradually penetrated into the inner surface of the adsorbent to adsorb at a relatively high pressure. Since MA has a uniform pore diameter, NH3 could easily penetrate into the inner surface, leading to an enhancement of the adsorption capacity. Although AC has the largest surface area among the 4 samples, its uptake capacity was smaller than MA and similar to Ze and Si samples. It was noted that the irregular pore structure of AC acted as a bottle-neck for the intrusion of NH3 molecules into the inner surface, and thus there was a decrease in the uptake of NH3. Although the Si sample had a similar surface area with MA, induced by the formation of aggregates between single silica nanoparticles, Si also showed a low uptake capacity. These results showed that the surface area of adsorbents did not significantly affect the adsorption capacity. A common feature of the AC and Si is their irregular pore size distribution. Namely, ill-defined pore structure reduces the adsorption of NH3 to adsorbents. Therefore, the surface area of the adsorbent is likely to be large, but the pore structure (i.e., regular interlinked pore system) is the most important factor for the NH3 adsorption.
The maximum uptake capacities (Qm, mg/g) for NH3 was calculated with Langmuir isotherm. As summarized in Table 2, the Qm values of the NH3 uptake on MA, AC, Ze, and Si adsorbents was 214.8, 45.8, 43.3, and 33.0 mg/g, respectively. MA had 4.7 times high uptake capacity for NH3, compared to AC. It was reported that commercial AC and zeolite have adsorption capacities for NH3 in the range 37–144 mg/g [7]. Therefore, MA used here is referred as a good gas-adsorbent with a very high adsorption capacity for NH3. The adsorption density (, mg/m2) of NH3 on the active surface of adsorbents was calculated using the well-known equation, VΔC/mS, where S, m, V, and C are parameters for the surface area, the amount of adsorbent, volume and adsorbed concentration of NH3, respectively [16]. The maximum adsorption density (Γmax) was obtained from the adsorption isotherm, and a similar trend with the maximum uptake of NH3 was observed (Table 2).
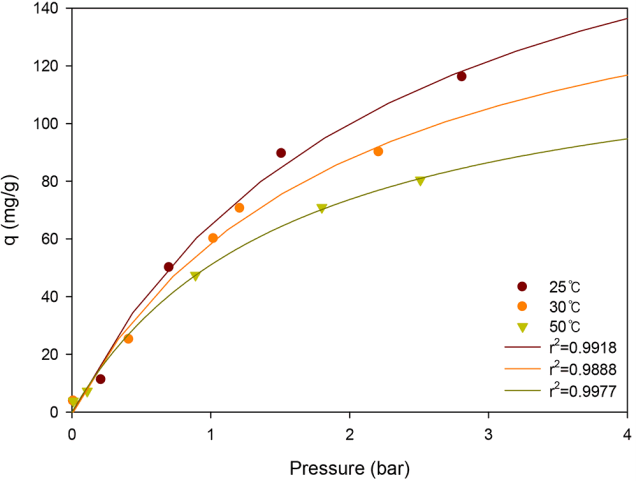
Because the adsorption of gas is strongly affected by temperature, the Gibbs energy and the uptake capacity for MA adsorbent were evaluated at different temperatures (25, 30, and 50°C). As shown in Fig. 5, the adsorption curve at high temperature was located below that of the room temperature. The maximum capacities of NH3 at 30 and 50°C were 174.5 and 132.6 mg/g, respectively, which were lower than those obtained at 25°C. With increasing temperature, -ΔG was slightly decreased from 12.0 to 11.7 kJ/mol, decreasing the interaction between NH3 and supports. Therefore, the adsorption system under high temperature poses a negative impact on the adsorption capacity, decreasing the amount of adsorbed gas.
Chemisorption and physisorption of NH3 on oxide material have been studied [12]. In the past literatures, it is reported that NH3 was chemisorbed on silica surface forming the NH3-O-Si [19] or NH2-Si [20] with a siloxane bond (Si-O-Si). For physisorption of NH3, the hydroxyl group was the preferred site to bond NH3 (NH3-OH-Si) [21]. Although the walls of calcined oxide (MS and MA) are made of multiple oxide bridges (Si-O-Si or Al-O-Al bonds), the pore surfaces contain free hydroxyl groups. These free polar groups serve as useful sites for adsorption of polar gas, such as NH3 [22]. In addition, the additional hydroxyl groups formed by hydrothermal conditioning with water increase the acidity of oxide materials, which results in an increase in NH3 capacity [5]. Therefore, similar to the investigation of NH3 adsorption on the silica surface, alumina (MA) has free hydroxyl groups, serving as a useful adsorption site for NH3. As calculated in our previous work [16], the moles of total surface sites per gram of solid were estimated to be 2.5 mol/kg for MA, assuming 5 hydroxyls/nm2 [23]. The maximum capacity of MA was higher than the total moles of hydroxyl sites (as described in isotherm results), and thus, NH3 adsorbed on the inner and outer surface of MA via both physisorption and chemisorption using hydroxyl groups and oxo-bridge.
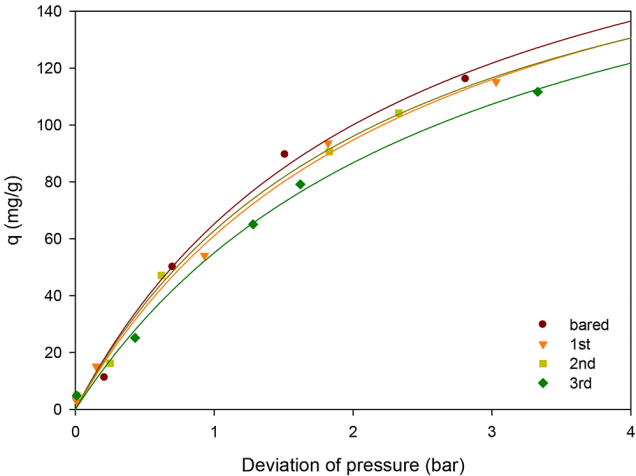
In field applications of NH3 adsorbents, maintaining the initial uptake capacity after regeneration is also an important factor for ideal adsorbents. After the regeneration, the feasibility of the adsorption–desorption cyclic operation was examined to investigate the reproducibility of MA performance, as shown in Fig. 6. The amount of adsorbed NH3 decreased slightly with repeated use of adsorbent (214.8 to 204.2 mg/g after 3 times recycling). While > 95% of the initial uptake capacity of NH3 was maintained after three cycles of operations, notably MA could undergo repeated adsorption–desorption cycles without any loss of activity.
4. Conclusions
MA was prepared by a post-hydrolysis method to use as the gas adsorbent for NH3. The MA adsorbent showed pore properties of ideal gas-adsorbents, such as uniform pore size, regular pore size distribution, interlinked sponge-like pore system, and large surface area. The NH3 adsorption performance of MA was compared to commercial gas-adsorbents (activated carbon, zeolite, and silica nanoparticle). This study on the isotherm adsorption represented a typical pressure-dependence as Langmuir isotherm. The maximum uptake capacity of NH3 for MA was 4.7–6.5 times larger than the same parameter for commercial adsorbents. There was no correlation between the uptake capacity and pore properties of adsorbents. Namely, the adsorption performance was not good, even if the surface area was large, as in the case of AC. A uniform pore structure with well-defined channels has been shown to more advantageous over a disordered pore network in terms of access to guest species to the binding sites [23]. Consequently, a regular pore structure (prefer to interlinked pore system) was found to be the most critical factor for the removal of NH3 using MA. In addition, because the adsorption energies of the toxic gases and common adsorbates followed the trend NH3 > PH3 > H2S > SO2 > CO (Barea et al., 2014), it was possible to apply as-prepared MA readily as conventional gas-adsorbents for H2S, PH3, and NH3.