1. Introduction
Oil spillage posed a global issue that has been occurring since the discovery of crude oil. Oil spill accidents often take place during oil utilization processes, resulting in energy loss as well as threats to the environment [1]. Oil transportation poses a highly risky business and oil spills need swift attention. As a matter of necessity, after an oil spill, marine cleanup operations need to collect or adsorb quickly a major part of any oil spilled [2].
Oil spills could be from human mistakes and carelessness, as well as deliberate acts of vandalism, war, illegal dumping, or by natural disasters such as hurricanes or earthquakes in some cases [3]. Massive marine oil spills have resulted in a great deal of damage to marine habitations. It had also led to economical impacts on fisheries, agriculture, tourism, and loss of energy source. Ayotamuno et al. [4] in 2005 reported that soils soaked with crude oil instantly loses fertility and initiates environmental degradation processes. Also, Broder and Zeller [5] in 2010 came up with the fact that oil spill had resulted in changes of soil pH which affected solubility and abundance of micro-nutrients. Inland water bodies are also affected by leaking of oil through pipelines, refineries, storage facilities, runoff from oil fields, and in some cases, process effluent from petroleum refineries and petrochemical plants. The methods commonly used to remove spill involve oil booms, dispersants, skimmers, in situ burning and so on. However, most of them require substantial financial input and their use is restricted because of cost factors, which often overrides the importance of pollution control [6]. Also most of the dispersants are often inflammable and cause health hazards to the operators and potential damage to animal and plant life. It could also lead to fouling of shorelines and contamination of water source.
Adsorption is identified as a cost-effective method to reduce the environmental problems of oil spill and pollution cleanups. The use of sorbent poses one of the most economical and efficient methods for combating oil spills. The accepted sorbent materials are affordable, accessible, have fast oil sorption rate, high oil sorption capacity (exhibits oleophilicity or lipophilicity), low water pickup, high oil retention capacity for transfer, high recovery rate of the adsorbed oil with easy methods, sound reusability, as well as high buoyancy properties.
Three major classes of oil sorbents, include organic synthetic products, inorganic mineral products, and organic natural products. Majority of the commercially available oil sorbents are organic synthetic products such as polypropylene (PP) and polyurethane materials.
Some of the mineral products used as oil sorbents include perlite, exfoliated graphite, vermiculites, organoclay, and zeolite to mention a few. Most of them usually have poor buoyancy and oil sorption capacity in the field. They could also be difficult to handle on site due to their granular or powder forms [1]. Most of them also exhibit poor oil recovery and reusability. As a result of their low hydrophobicity, they may also experience collapse of their microstructure due to water sorption. Exfoliated graphite and silica gel seems to be excellent oil sorbents, but they are fairly expensive. These limitations have led to the recent interest in seeking alternative materials, especially biodegradable natural agro-based products.
An agricultural solid waste seems cheap, abundant, and easily available. Over the years, some researchers have focused on the feasibility of the low-cost materials that were derived from agro-wastes for oil and heavy metals removal [7]. Effective usage of these waste materials enhances their reuse and savings in disposal fee. As a supportive means of enhancing the quality of this present work, previous contributions in the field of adsorption and waste water treatments were acknowledged [8–23].
In this research, mango shell (MS) and mango shell activated carbon (MSAC) were used as the adsorbent, without the edible part of the peel or fibrous material. The seeds are within the nuts, and are not the consumable part of mango, usually discarded as wastes. Activated carbon prepared from MS has been applied for colour adsorption and removal of heavy metals from waste water due to its well developed pore volume and large surface area [24]. Some researchers have converted mango seed into activated carbon [25], but more investigation into the usage of MS and MSAC in adsorbing crude oil seems relatively few to other reported agro wastes.
The objective of this study is therefore on the possibility of using an agro-waste like MS and MSAC to adsorb crude oil, investigating various relevant physical parameters as they influence the adsorption process. The usage of Langmuir, Freundlich, Temkin and Dubinin Radushkrvich isotherm models is employed to evaluate the sorption capacity of the adsorbents as well as to determine the kinetics and optimum parameters under which adsorption of crude oil with the two adsorbents are most effective. Statistical application of Analysis of Variance (ANOVA) and Bonferroni-Holm Posthoc significance test to justify the adsorption operation is also examined. The outcome of the research is expected to propose MS and MSAC as useful materials for environmental control of crude oil spills.
2. Materials and Methods
2.1. Apparatus and Reagent
The following materials and apparatus were utilized for the experimental work:
England Pyrex beakers (50–500 mL), England Pyrex conical flasks (50–500 mL), England Pyrex measuring cylinders (50–1,000 mL), England Surgifield Model SM 1002A Muffle electric furnace, Parsippany, New Jersey, United States OHAUS Model P4102 Electronic Mass Balance, Germany Merck KGaA Whatman (Model 1001–125) 125 mm diameter filter paper, England Mesh Sieve (Model BSS 410/43), Lemfield Medical England Model DHG9053A Heating Drying Oven, United States Hanna Instruments model HI98107 pH meter, spatula, United States The Lab Depot Inc. Model RS232 orbital shaker, Australia RATEK model SWB20D water bath shaker, United Kingdom Jenway Model 6405 Ultraviolet (UV) spectrophotometer, de-ionized water, Nigerian Niger Delta Bonny Light crude oil, MS, 85% w/w ortho-phosphoric acid and United States PerkinElmer Fourier Transform Infrared (FTIR) Spectrophotometer Spectrum 2000 Model.
2.2. Preparation of Simulated oil Spill
A typical small oil spill of 0.025 g/L initial concentration was simulated by pouring 25 g of crude oil into 1,000 mL of distilled water in a beaker.
2.3. Preparation of Adsorbent
The mango nuts collected were washed and dried in the sun at room temperature for one week and cracked using hammer, the shell was then removed and dried under sun light at room temperature for two weeks. The shell was oven-dried at 105°C after which they were crushed using laboratory mortar and pestle. The particles were sieved with 80 μm sieve size. Some part was stored in a plastic bottle for experimentation. Some other sieved samples were impregnated in H3PO4 at impregnation ratio of 1:1 by measuring 100 g of sample in 100 mL of reagent. The impregnated sample was left at room temperature for 24 h. After impregnation, the excess solution was filtered off. Finally the resulting sample was carbonized in a heated furnace at a temperature of 450°C for 30 min. The activated carbon was rinsed with distilled water and dried at 105°C for 1 h in the furnace. Finally the activated carbon produced was kept in a tightly closed plastic bottle.
2.4. Characterization of Adsorbent
FTIR spectroscopic analysis was performed on the MS and MSAC using FTIR spectroscope (FTIR-2000, Perkin-Elmer) to determine the functional groups present. The spectra were measured from below 400 to 5,000 cm−1.
2.5. Batch Adsorption Equilibrium Studies
Exactly 100 mL of the simulated oil spill of initial concentration of 0.025 g/L (CO) was used for each study. The equilibrium concentration of crude oil in water for each measured sample was determined using UV spectrophotometer. The weight (m) of the adsorbents, volume (V) of simulated oil spill and the corresponding equilibrium concentration (Ce) were recorded. For each batch run, the quantity of crude oil adsorbed per unit weight of adsorbent denoted as qe was determined with Eq. (1) expressed by Uzoije et al. [26] as:
The percent removal is expressed in Eq. (2) as:
The mean values were calculated and utilized after double runs for reproducibility errors.
2.5.1. Effect of adsorbent dose
About 0.025 g/L initial concentration oil sample was added to mixtures of 1.0 g, 2.0 g, 3.0 g, 4.0 g and 5.0 g of 80 μm MSAC, respectively in a 500 mL beaker. The beaker was placed on an orbital stirrer, and then stirred at 200 rpm at temperature of 25°C for an hour each. The absorbance values was recorded carefully during the experiments, this was also repeated for the raw mango sample.
2.5.2. Effect of contact time
About 0.025 g/L initial concentration oil sample was added to a mixture of 2.0 g of 80 μm MSAC in a 500 mL beaker. The beaker was placed on an orbital stirrer, and then stirred at 200 rpm for various contact time: 15, 30, 45, 60, and 75 min. After stirring, the samples were withdrawn at the specified time intervals, filtered through a filter paper and the filtrate measured using a UV machine. The absorbance values were then recorded; this was also repeated for the raw mango sample.
2.5.3. Effect of temperature
Batch adsorption experiments were carried out at different temperatures of 15, 25, 35, 45 and 60°C. About 2.0 g of the MSAC was shaken with 0.025 g/L initial concentration oil sample in a 500 mL beaker at 200 rpm for 1 h at various temperatures. The wetted adsorbent was then removed from the beaker, while the oil and water filtered through a filter paper and the filtrate measured using a UV spectrophotometer. The absorbance values were obtained and recorded. This was also done for ordinary mango samples.
2.5.4. Effect of pH
The effect of pH on crude oil adsorption was studied by agitating 2 g of the activated carbon in a 200 mL of the prepared samples with concentration of 0.025 mg/L at different pH values, which ranged from 5–9. The samples were agitated for 1 h to reach equilibrium and agitation speed was maintained at 200 rpm. The pH of the solution was adjusted to the desired value by drop wise addition of 0.1M hydrochloric acid (HCl) and 0.1M sodium hydroxide (NaOH). After removing the adsorbent, the oil and water were filtered through a filter paper and the filtrate measured using a UV spectrophotometer. The absorbance values were recorded. This was also repeated for ordinary mango samples.
2.5.5. Effect of adsorbate dose
Batch adsorption experiments was carried out at different adsorbate dose in which 2.0 g of MSAC was shaken with 2, 4, 6, 8, and 10 g crude oil samples per 200 mL water mix in a 500 mL beaker to give varying oil concentrations at 200 rpm for 60 min using constant ambient temperature. The wetted adsorbent was then removed from the beaker, while the oil and water were filtered through a filter paper and the filtrate measured using a UV machine. The absorbance values were then taken. This was repeated for raw mango samples.
2.5.6. Effect of rotational speed
Batch experiment was carried out by adding 0.025 g/L initial concentration oil sample to a 2.0 g 80 μm corncobs adsorbent in a 500 mL beaker. The beaker was placed on an orbital stirrer, and then stirred at constant room temperature for a specified revolution per minute for 60 min. Various rotational speeds of 100, 200, 300, 400 and 500 rpm were used. After stirring, the samples were withdrawn at the specified time intervals, filtered through a filter paper and the filtrate were measured using a UV machine. The absorbance values were recorded. The procedure was repeated for raw mango samples.
3. Results and Discussion
3.1. Physical Properties of Adsorbents
Some physical properties of the adsorbents were determined. Typical average values of the percent moisture content, bulk density, specific gravity and porosity were 1.2%, 0.42 g/mL, 0.57 and 0.69 for MS, while 1.8%, 0.40 g/mL, 0.70 and 0.92 were obtained for MSAC, respectively.
3.2. Fourier Infra-Red Spectroscopy Images of Adsorbents
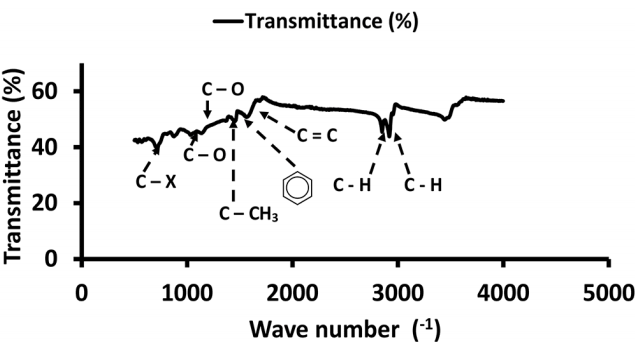
Fourier Infra-Red spectroscopy conducted on the MS and MSAC revealed the presence of many peaks indicating different functional groups as presented in Fig. 1 to 4 below, which make the adsorbents viable for adsorption. All assignments were made and observed in accordance with Coates [27]. The heavier functional groups could be identified at the smaller wave numbers, while the lighter aliphatic bonds sited at the larger wave numbers.
The FTIR analysis revealed the presence of many peaks indicating various functional groups within the adsorption operation. The spectrum of the MS before and after adsorption showed the broad and intense peak between 2,851 cm−1 and 2,953 cm−1 which was attributed to the stretching of C-H group due to alkyl bond. The peak also observed at 1,242.99 cm−1 was associated with stretching vibration of C-O group due to carboxylic acid. The peak around 1,500 cm−1 corresponded to the C-C stretching which might be attributed to aromatic bond.
The FTIR spectrum of MSAC before and after adsorption showed the peak between 2,840.20 cm−1 and 2,901.04 cm−1 was attributed to the stretching of C-H group due to alkyl bond, and that observed at 1,451 cm−1 was due to C-C stretching which might be attributed to aromatic bond.
This showed that the adsorption process caused the peak fall in transmittance which indicated that some functional groups were present.
3.3. Batch Adsorption Equilibrium Studies Results
3.3.1. Effect of adsorbent dose
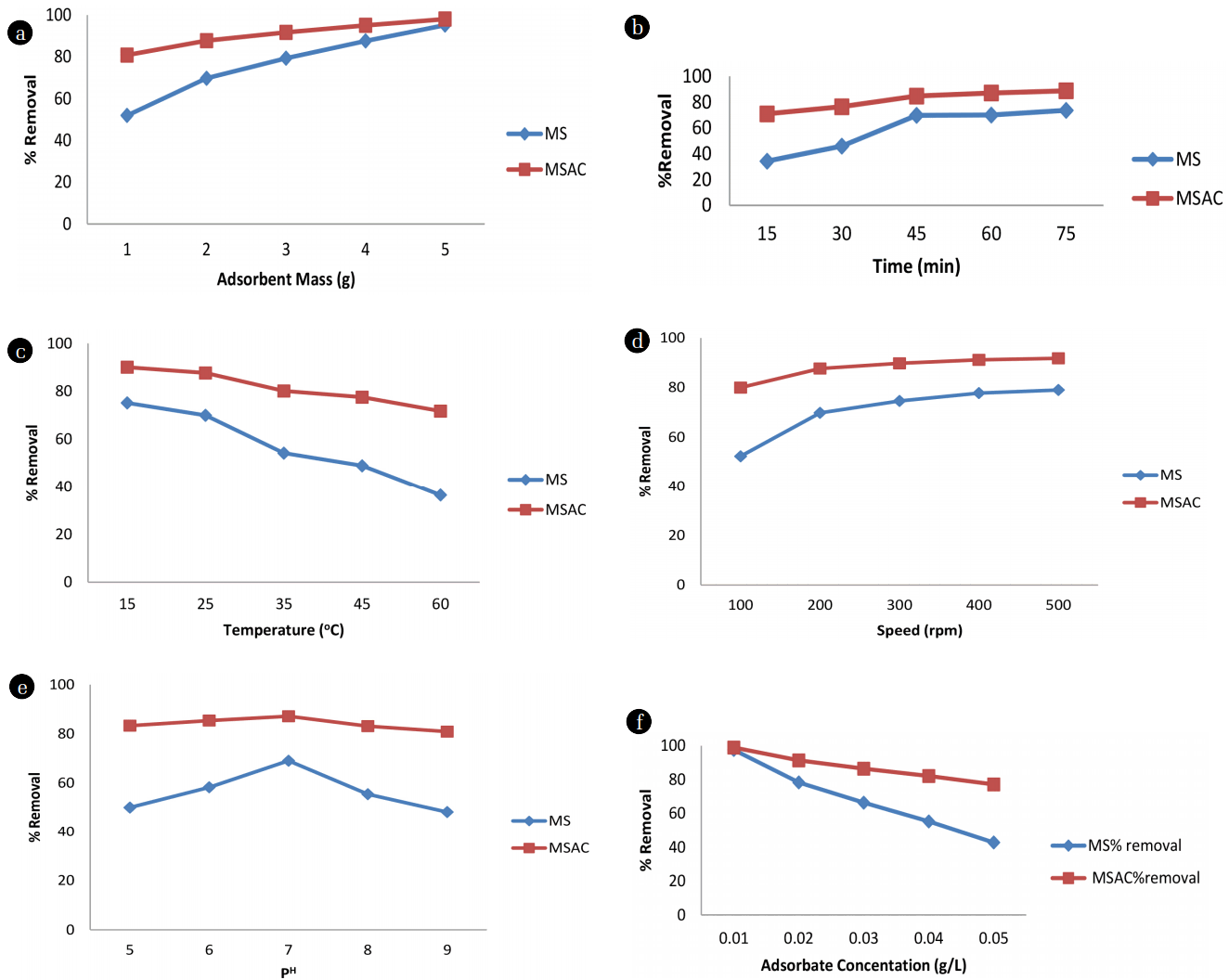
It was observed from Fig. 5(a) that as the adsorbent increased from 1 to 5 g, the percentage removal increased. The increase in the adsorption percentage with increase in dose of adsorbent was due to the increase in active sites of the adsorbent and thus making easier penetration of the crude oil molecules into the adsorption sites. Similar experience was observed by Itodo and Itodo [28].
3.3.2. Effect of contact time
The extent of crude oil removal by the two adsorbents was found to increase as the contact time increased from 15 to 75 min as shown in Fig. 5(b). The adsorption of crude oil using the adsorbents was found to be rapid initially and then become slower with the increase of contact time. This may be due to the reducing strong attractive forces between the crude oil molecules and the adsorbent as contact time increased especially after sometime between 60 to 75 min. It gradually approached almost constant value at equilibrium. The percentage removal of MSAC was higher than that of the inactivated MS which also showed that more active sites were available in the MSAC.
3.3.3. Effect of temperature
The batch adsorption experiments result carried out for different temperatures of adsorbent shown in Fig. 5(c) indicated that adsorption capacity was higher at lower temperatures. The percentage removal was high at 15°C for the temperature range considered. The results further showed that the percentage removal of the crude oil decreased with increase in temperature, indicating the dependence of adsorption on temperature similarly observed by Bello and Ahmad [29]. The percentage removal of the MSAC was also higher compared to that of the ordinary MS which showed that the MSAC possessed more energy required for adsorption.
3.3.4. Effect of mixing speed
The batch adsorption experiment result at different mixing speed as shown in Fig. 5(d) revealed that the adsorption of crude oil onto the adsorbents increased with speed. Lin and Liu [30] reported that mass transfer rate increases with increase in stirring speed. The increase in stirring speed resulted in a reduction in surface film resistance, thereby allowing residual oil to reach the particle surface more easily. Surface film resistance hinders rate of adsorption. The result showed that the rate of adsorption with oil removal gradually increased with speed and the most remarkable adsorption occurred at about 500 rpm similar to the work of Olufemi et al. [31].
3.3.5. Effect of pH
Surface charges on the adsorbent and degree of ionization of the adsorbate could be influenced by the pH of the solution, which makes pH an important factor in adsorptive studies. The batch experiment result shown in Fig. 5(e) indicated that varying the pH using the presence of either OH− or H+ ions compete with molecules of crude oil for adsorption site. The higher or lower the pH, the higher the concentration of either ion and thus the affects uptake of crude oil molecules. For both adsorbents the neutral pH seems to be more favourable.
3.3.6. Effect of adsorbate concentration
The effect of adsorbate concentration is shown in Fig. 5(f) which revealed that adsorption of crude oil decreased with increase in adsorbate concentration for both adsorbents. Basically this can be explained in terms of available active sites. At low adsorbate concentration, the ratio of surface active sites to crude oil was high. Hence the crude oil interacted with the sorbent to occupy the active sites on the carbon surface sufficiently, and was removed from the solution as observed in some adsorption processes [32, 33], but with the increase in adsorbate concentration, the number of active adsorption sites was not enough to accommodate crude oil molecules.
3.4. Adsorption Isotherms
The Freundlich, Langmuir, Temkin and Dubinin-Radushkevich isotherms were used to investigate sorption processes. They are expressed in Eq. (3) to Eq. (6) as follows:
A linear form of the Freundlich expression is:
The Langmuir Isotherm is given as:
The linearized Temkin Isotherm is expressed as:
The linearized Dubinin-Radushkevich Isotherm is expressed as:
In Eq. (6), BD is closely related to the free energy of sorption per mole of the sorbate as it moves to the surface of the adsorbent from infinite distance in the solution and qD is a constant related to the degree of sorbate sorption by the adsorbent surface.
The values of the isotherms constants under various operating parameters for MS and MSAC are given in Table 1. Comparing the correlation coefficient (R2) values after linearizing and plotting the linear straight line relationships for all the isotherms, indicated that the Temkin isotherm favored the two adsorbents better than other isotherms. This confirmed the relative importance of temperature in the energy involved for the sorption processes.
3.5. Optimization
A Statistical Design of Experiment MINITAB © software was used to predict the optimum values of percentage removal of each parameter on the overall process. A full factorial design of experiment was used, and afterwards percentage removal was calculated. All the parameters were selected as well as factor levels coded as -1 (low), 0 (middle) and 1 (high).The full factorials design gave 54 runs of experiment and the optimum values obtained are given in Table 2 for MS and MSAC.
3.6. Statistical Analysis
The ANOVA and Bonferroni-Holm Posthoc significance test for crude oil adsorption by MS and MSAC with varying parameters are given in Table 3.
Fisher’s F is a ratio of the variance between groups to the variance within groups and P is a probability factor that must be less than 0.05 using the 95% confidence interval in the ANOVA analysis. The degree of freedom between the groups was 1, while the degree of freedom within the groups was 8 for each adsorbent.
For MS, the F and P values implied a strong dependence of Ce on the various parameters investigated (CA, M, t, T, r and pH). The Bonferroni-Holm Posthoc parametric significance test for various parameters investigated showed very strong dependence or significance on each other except for the relationship between Ce and M with a P value of 0.198883, which was greater than 0.05 in the 95% confidence interval.
This could be that the mass of adsorbent used to contact the adsorbate was too much than the relevant quantity needed, hence seems not to constitute a limiting factor in the adsorption process.
In the case for MSAC, the ANOVA and Bonferroni-Holm Posthoc significance test for crude oil adsorption with varying parameters as also given in Table 3 showed the actual values of the sum of squares within the groups and the sum of squares between the groups. There existed a statistically wide variation between the individually investigated parameters and the equilibrium concentration groups at the 95% confidence interval, as the F (1, 8) and P values are mostly within expected ranges. Fisher’s F is still the ratio of the variance between groups to the variance within groups and P is a probability factor that must be less than 0.05 in the ANOVA analysis. The degree of freedom between the groups was also 1, while the degree of freedom within the groups was still 8. The F and P values implied a strong dependence of Ce on the various parameters investigated (CA, M, t, T, r and pH).
The Bonferroni-Holm Posthoc parametric significance test for various parameters investigated showed very strong dependence or significance on each other except also for the relationship between Ce and M with a P value of 0.537811, which was greater than 0.05 in the 95% confidence interval. This could also be interpreted to mean that the mass of adsorbent used to contact the adsorbate was too much than the relevant quantity needed, hence seems not to constitute a limiting factor in the adsorption process.
3.7. Kinetic Studies
The pseudo first order kinetic model is given in Eq. (7) as:
The pseudo second order kinetic model is given in Eq. (8) as:
The constants from the plots of the pseudo first and second order kinetics are given in Table 4. It can be seen that the pseudo first order kinetics gave a better fit for the two adsorbents with values of 0.943 and 0.872 for MSAC and MS, respectively.
4. Conclusions
The possibility of mango shell as a natural adsorbent to remove crude oil had been investigated in this work. The results demonstrated that crude oil removal by adsorption onto MS being a waste material is feasible. The adsorption and percentage removal of crude oil was found to be dependent at various levels on adsorbent dose, adsorbate dose, contact time, temperature and pH as well as speed of agitation. The adsorbed oil-water ratio of the mango adsorbent was more than unity in all cases investigated, which indicated higher adsorption selectivity for crude oil over water by the adsorbent. The considered adsorption temperature range was between 15 to 60°C, with the maximum adsorption observed at 15°C.
The correlation coefficient indicated various dependencies on parameters utilized by the adsorption process. Based on the high affinity for oil with low water pick up, MS used as an adsorbent could be expressly regarded as an oleophilic or hydrophobic substance. The equilibrium adsorption isotherm analysis with Langmuir, Freundlich, Temkin and Dubinin Raduskevich presented fitted the equilibrium data reasonably well, but Dubinin Raduskevich isotherm seems least significant because of the low R2 values. In conclusion, the result from the Temkin isotherm model seems to be the most significant, similar to the findings of Sidik et al. [34]. Optimum parametric values needed for the adsorption process were also presented. The results obtained with the Bonferroni-Holm Posthoc parametric significance test showed that the lowest P value for MS was 0.003261, while that for Mango shell activated carbon was 0.002991 under the same condition of variation of equilibrium concentration with mixing speed. The enhanced effect brought about by activation of the mango shell to provide an adsorbent with higher surface area and capacity for adsorption was therefore justified statistically using ANOVA and Bonferroni-Holm Posthoc significance test. The pseudo first order kinetics gave a better fit for the two adsorbents used for the adsorption operation.
Nomenclature
AT
Temkin Isotherm Equilibrium Binding Constant (dm3/mol)
BD
Dubinin – Radushkevish isotherm energy parameter
bT
Temkin Isotherm Constant (J/mol)
CA
Adsorbate Concentration, mg/L
Ce
Equilibrium Concentration of Mixture (mg/L)
C0
Initial Concentration of Mixture (mg/L)
DFB
Degrees of freedom between groups
DFW
Degrees of freedom within groups
F
Fisher’s ratio
h
Pseudo second order constant (gmg−1min−1)
K1
Pseudo First Order Constants (min-1)
Kad
Dubinin–Radushkevich Isotherm Constant (mol2/kJ2)
KF
Freundlich equilibrium constant which indicate adsorptive capacity
KL
Langmuir Constants (mg/g)
M, m
Mass of the Adsorbent (g)
n
Freundlich constant which indicate adsorptive capacity
P
probability factor
PC
Critical probability factor
pH
Level of acidity or alkalinity
qD
Dubinin-Radushkevich isotherm constant
qe
Amount of Solute Removed or Adsorbed at Equilibrium (mg/g)
qm
Theoretical Isotherm Saturation Capacity (mg/g)
qs
Theoretical Isotherm Saturation Capacity (mg/g)
qt
Amount of Solute Removed or Adsorbed at any time (mg/g)
r
Mixing speed, rpm
R
Universal Gas Constant (J/mol K)
R2
Coefficient of Regression
SSB
Sum of squares between groups
SSW
Sum of squares within groups
t
Mixing time, min
T
Temperature, K
V
Volume of Simulated Oil spill (L)