1. Introduction
Increasing the demand for pork resulted in the generation of enormous amounts of swine wastewater. Swine wastewater is composed of feces, urines and washing water discharged from pig farm contains high nitrogen content (total nitrogen; TN, 1.8–4.3 g/L) and organic matter content (total chemical oxygen demand; TCOD, > 20 g/L) [1]. Excess discharge of nitrogen has not only led to considerable eutrophication, but has also caused loss of nitrogen resource. Biological denitrification process is accepted as traditional and economical method to remove nitrogen in the wastewater. However, high concentration of nitrogen decreases performance of the biological process because the activity of micro-organisms in biological treatment system depends on proper carbon-to-nitrogen (C/N) ratio [2]. Lower the concentration of ammonia prior to the biological treatment process is necessary.
Large amounts of ammonium nitrogen have a high potential for product as an ammonium based fertilizer. Recovery of resource from wastewater is required for a more sustainable future. Several physicochemical process such as struvite precipitation [3], ammonia stripping [4], electrodialysis [5], reverse osmosis [6] and microwave (MW) radiation [7] have been proposed to remove and recover ammonia nitrogen in the wastewater. Struvite precipitation needs additional reagents, which may introduce new pollutants to water body. Ammonia stripping makes use of stripping tower and consumes much energy. Electrodialysis and reverse osmosis technology are required to periodical membrane exchange because of membrane fouling [6].
In recent years, MW radiation has attracted a great deal of attention for application of wastewater due to possibility of high concentration of ammonia (> 1000 mg/L) [8], its high removal efficiency and reaction rate [8], controllable recovery rate [9], quick reaction time, easy control, and the yield and purity of products increasing [10]. MW radiation technique was applied to remove ammonia from municipal wastewater and coke-plant wastewater [7]. The pilot-scale application of MW radiation was successfully operated using coke-plant wastewater [8].
The heating and molecular moment generated by MW facilitate to evaporate volatile ammonia molecules in solution (Eq. (1)) [7]. Ammonia gas stripped is collected in acid solution such as sulfuric acid, so that ammonium sulfate is formed (Eq. (2)) [10]. Ammonium sulfate can be applied to alkaline soil as an inorganic nutrient supplement.
Currently, nitrogen recycling technology from variety of waste-water is receiving focus by the scientists and industrialists [11]. Only a few scientists reported microwave radiation as pretreatment of the raw wastewater [12], even no reports of microwave radiation from swine wastewater. This study investigates the optimum ammonia nitrogen removal condition and recovery of ammonia nitrogen from swine wastewater by microwave radiation.
2. Materials and Methods
2.1. Swine Wastewater
Swine wastewater was collected from the piggery waste storage (Jeongup, Korea) and refrigerated at −80°C before use. The compositions of the swine wastewater used in this study are shown in Table 1.
2.2. Microwave Reactor System Set-up and Operation
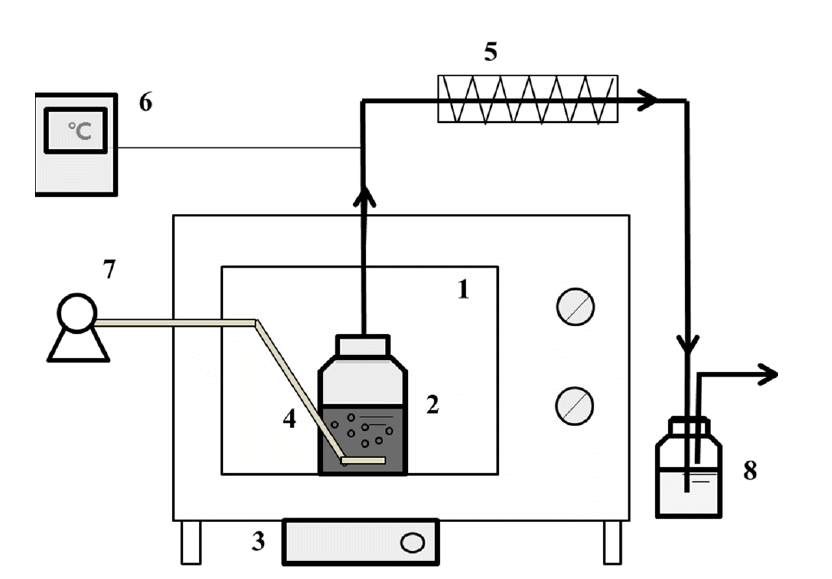
The schematic diagram of the microwave reactor system is shown in Fig. 1. The experimental apparatus of microwave radiation process was designed and set-up according to previous report [7]. A domestic microwave oven (KR-B20MB; Daewoo electronics, Korea) with multiple power settings was used as the source of the MW radiation. A controllable stirrer was installed at a bottom of the oven. A hole was drilled on the top of the oven for connection with condenser and reactor; 26 mm ports for a glass tube with thermocouple and condenser on the top of the oven. The hole was jacketed with copper tubing to avoid microwave leakage. The condenser was used to prevent evaporation and connected with 100 mL of the H2SO4 (0.5 mol L−1) to capture ammonia gas. A 150-mL bottle reactor with Pyrex was placed in the oven. A 50 mL of wastewater sample was filled in the bottle reactor. The initial pH of the solution was adjusted using NaOH (5.0 mol L−1). The MW radiation reaction was performed at different parameters and conditions: 1, 2, 3, 4, 5, and 6 minute of radiation time; 8, 9, 10, 11, and 12 of pH; 300, 500, and 700 Watt (W); 0, 0.25, and 0.5 L/min of aeration rate; 0, 300, and 600 rpm of rotation rate. Each experiment was conducted in triplicate.
2.3. Analyses
Ammonia nitrogen, chemical oxygen demand (COD), and pH were measured. The volume of samples was always adjusted with deionized water to maintain the same initial volume before measurement because the volume of solution slightly decreased by reason of evaporation of water after reaction. COD was measured by a spectrophotometric method (Humas Method 1002; Humas, Korea). The ammonia nitrogen was analyzed by Nessler method using water test kit (Humas Method 2811; Humas, Korea). pH was measured by pH meter (780 pH meter; Metrohm, Switzerland).
3. Results and Discussion
3.1. Optimization of Operation Parameters
3.1.1. Effect of Initial pH
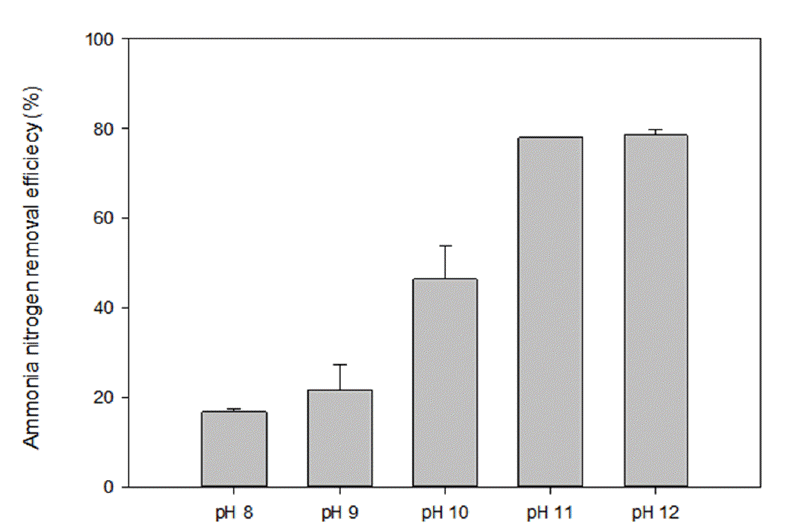
To investigate the effect of initial pH, batch experiments were performed by adjusting the pH of swine wastewater and were conducted at 700 W for 4 min. The influence of pH on the ammonia nitrogen removal by MW radiation is shown in Fig. 2. As the initial pH was increased, the ammonia nitrogen removal efficiency showed significantly an increase until pH 11 reached and similar efficiency between pH 11 (78.1%) and pH 12 (78.6%). It was found that the pH decreased around 1.0 after reaction in MW radiation system.
In acidic and neutral condition, most ammonia nitrogen is existed as NH4+. In basic condition, non-volatile NH4+ converts to volatile NH3. At high pH, ammonia volatilization is more facilitated by driving the equilibrium between NH3 and NH4+ to molecular ammonia [7]. It is therefore, the relationship of soluble ammonium ion (NH4+) and dissolved molecular ammonia (NH3) results in alkaline of optimal pH condition. Microwave radiation can be applicable to rapid heating by rotating polar molecules in waste-water fleetly which led to improvement of molecular movement and consequently facilitate to evaporate volatile NH3. Gas bubbles caused by boiling solution help molecular NH3 to strip from the wastewater. In addition, microwave radiation might contribute to decrease the activation energy and to make a weak molecular chemical bond [7]. Through the result and mechanism of nitrogen removal in microwave radiation process, it is necessary to adjust pH in order to reduce content of ammonia nitrogen by stripping volatile NH3.
The 98.2% of ammonia removal efficiency at pH 11 with aeration was attained by Lin et al [7]. Rabah and Darwish [13] also achieved pH 11 as optimal pH condition which resulted in 91% NH3-N removal. In this experiment, pH 11 was also selected as the optimal condition considering the removal efficiency and the minimal dosage of alkali added.
3.1.2. Effect of Radiation Time
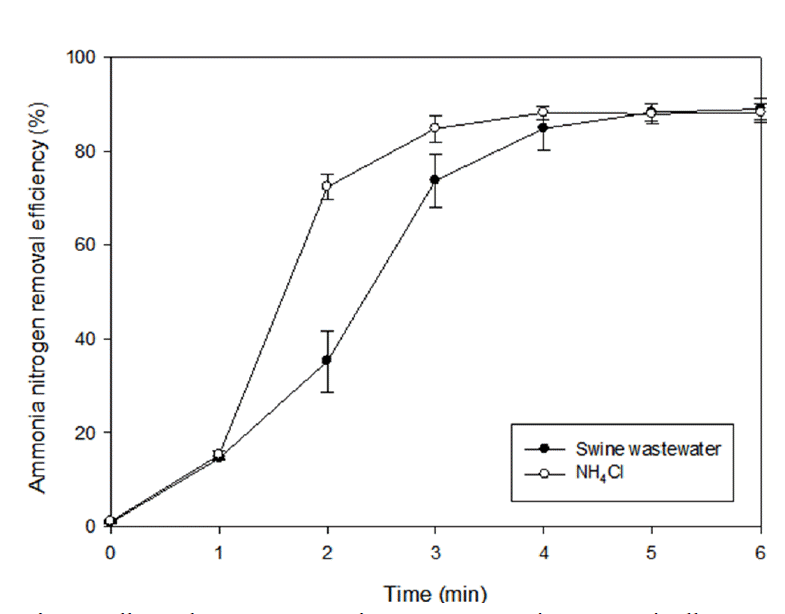
The effect of MW radiation time was investigated for efficient ammonia nitrogen removal from NH4Cl solution and swine waste-water at pH 11, 700 W. The concentration of ammonia nitrogen in the NH4Cl solution adjusted was similar to the concentration of ammonia nitrogen in the swine wastewater. Fig. 3 showed that the ammonia nitrogen removal efficiency increased with radiation time and reached a plateau after 4 min (NH4Cl solution) and 5 min (swine wastewater) of MW radiation, respectively. The ammonia nitrogen removal efficiencies of NH4Cl solution and swine wastewater were each 88.3% at 4 min and 88.2% at 5 min. Swine wastewater might be required more heat to evaporate same NH3 content than NH4Cl solution due to mixture effect which has a lot of component such as organic matter and other ions. The longer MW radiation time caused solution temperature to become higher which induced more active molecular motion. It facilitated the escape of ammonia nitrogen from solution. Because significant increment in ammonia removal efficiency was not showed and longer radiation time would cause excessive cost, 5 min was determined to be the optimal radiation time.
3.1.3. Effect of Power
The MW radiation reaction was performed using swine wastewater at different microwave power at pH 11. Fig. 4 (a) shows that the plateau of residual NH3-N concentration at the condition of 300, 500, and 700 W was attained after 5.0, 6.7, and 12.5 min, respectively. The selection of power value was related to the amount of energy consumption of MW radiation during the reaction. Fig. 4 (b) shows that the NH3-N removal efficiency is almost same regardless of output power relationship with quantity of the energy consumption. This result indicates that ammonia nitrogen removal efficiency depends on the amount of energy consumption considering reaction time and power value. The reaction time should be considered for efficient operation, so that 700 W value was determined to remove large volume of ammonia nitrogen quickly.
3.1.4. Effect of Aeration and Stirring
To identify effect of aeration and stirring on ammonia nitrogen removal in MW radiation system, the removal efficiency of ammonia at different aeration flow rate and stirring speed with 700 W was investigated. This experiment was conducted at pH 11 and 4 min. The increase of ammonia removal efficiency was less than 4% using the aeration and stirring method in Table 2. Both aeration and stirring might attribute to increase mass transfer of ammonia nitrogen in the solution due to turbulence and agitation [14]. However, it was observed that the whole solution was circulated well by boiling in the reactor during MW radiation even without aeration and stirring. It might be a reason that both aeration and stirring are not significant influenced on the ammonia removal. Lin et al. [7] also found similar result about aeration effect at pH 11. Ammonia removal efficiency was close between aeration and no aeration after MW radiation.
3.2. Ammonium Sulfate Recovery
To confirm the recovery rate in the microwave radiation process, NH3-N concentration in both effluents of the microwave radiation and absorption solution were measured after operation using swine wastewater at optimum condition (pH 11, 5 min, 700 W). The initial ammonia nitrogen concentration was 2,494 mg L−1 in swine wastewater. The ammonia stripped from swine wastewater was collected in H2SO4 solution. The ammonia removal efficiency was 88.9% and the 82.6% of initial ammonia nitrogen content was recovered as ammonium sulfate. The loss of nitrogen was probably due to two reasons; one is ammonia converted into other nitrogen form, another is stripped ammonia stayed in the reactor system such as connecting tube or condenser. To identify whether ammonia convert into nitrate and nitrite or not, content of nitrate and nitrite ions were also measured. There were slightly decrease or no difference between the initial and final samples of swine waste-water after MW radiation reaction. Therefore, it would be more reasonable second possibility. This result could be demonstrated that the content of NH3-N in swine wastewater was reduced by volatilization mechanism; ammonium ion was converted into volatile ammonia (free ammonia) in basic solution and then stripped [7].
3.3. Effect after MW Radiation
The SCOD value increased slightly from 10,883 mg L−1 to 11,639 mg L−1. TCOD value decreased about 3% of initial concentration through the MW radiation. Increasing level of soluble organic matter was related to decreasing level of the TCOD. Microwave radiation was used to treat sludge because the MW radiation makes chemical bond break easily by releasing of soluble organic components into the water phase [15]. The SCOD/NH3-N ratio of MW radiation process increased from 6.8 to 49.4. It might help to improve efficiency of subsequent biological treatment such as anaerobic digestion because of proper C/N ratio to microorganisms in the biological process. The degradation of performance in anaerobic treatment caused by unbalance of C/N ratio has been known [16].
4. Conclusions
In this work, the performance of the MW radiation process was evaluated from removal efficiency of ammonia nitrogen in the swine wastewater at different pH, radiation time, power, aeration, and stirring conditions mainly. It was found that pH, radiation time, and power value are significantly influenced on the removal of ammonia nitrogen, while aeration and stirring showed minor effect of the removal ammonia nitrogen. Ammonia nitrogen removal efficiency from power value depends on the amount of energy consumption considering reaction time. The 88.9% of ammonia nitrogen in the real swine wastewater was removed and 82.6% of initial ammonia nitrogen content was recovered as ammonium sulfate at optimized conditions (pH 11, 5 min, 700 W). MW radiation can be considered as alternative technique to remove and recover high concentration of ammonia nitrogen in the swine wastewater.