1. Introduction
Post biological denitrification is applied to a variety of environmental areas, such as groundwater remediation, water treatment, and wastewater reclamation [1, 2]. Technologies related to biological denitrification could be divided into heterotrophic and autotrophic denitrification in terms of the microorganisms involved and carbon source utilized. Heterotrophic denitrification requiring external carbon source is the most common and proven due to its high kinetic performance and stability. However, the addition of an external carbon source for post-denitrification may lead to an increase in operation cost and cause problems, such as excess biological growth and residual organic carbon in the treated effluent. Autotrophic denitrification using elemental sulphur or hydrogen gas can be an option for heterotrophic denitrification with several advantages: less sludge production and no external carbon source required [3–5]. Nonetheless, intermediate sulphate occurrences and high operating cost for the use of hydrogen gas can be addressed as major defects of autotrophic denitrification.
Denitrification using methane as a sole carbon source is already known [6] and application of this to water treatment has recently been attempted [7]. Since methanotrophic reaction converting methane to methanol is most likely to occur in the presence of oxygen, methanotrophic denitrification has so far been known as aerobic oxidation of methane with denitrification (AeOM-D). The methanotrophs utilize methane monooxygenase (MMO) to oxidize methane to methanol (or other organic carbon) and these groups are currently classified into three specific types depending on the metabolic pathway: type I (ribulose monophosphate [RuMP] pathway), type II (serine pathway), and type X (simultaneous RuMP and serine pathway) [8]. The stoichiometry for AeOM-D is shown in Eqs. (1) and (2).
Meanwhile, more recent studies have confirmed that methane can be directly used as a carbon source for denitrification in the absence of oxygen, that is, anaerobic oxidation of methane with denitrification (AOM-D) [8, 9]. The stoichiometry for AOM-D is shown in Eq. (3) and the reaction is thermodynamically favoured [10].
AOM-D has several advantages over AeOM-D, when applied for water treatment: 1) it does not require oxygen (or air) supply; 2) it theoretically needs 25% less CH4 for denitrification; and 3) it does not generate by-products, such as methanol, which may cause deterioration of effluent. However, few studies have been successfully done due to the limited rate for denitrification and slow growth of the related methanotrophs [11, 12]. Pure cultures have not yet been isolated; however, several studies presented that anaerobic methane oxidation can be conducted by anaerobic methanotrophic archaea [13, 14].
Since AOM-D has such drawbacks in denitrification rate and methanotrophic growth, it is of importance in enhancing kinetics for the mass transfer of methane. Mass transfer of methane to water is very low due to its high dimensionless Henry’s constant (28.4 at 20°C). Therefore, exposure of methanotrophs to abundant methane will be a key factor to enhance the AOM-D performance. Islas-Lima et al. [15] confirmed the capability of AOM-D in anaerobic batch assays, through varying methane partial pressures in the headspace. The nitrate removal efficiency reached 99.3% regardless of the methane partial pressure; but the denitrification rate was dependent on the methane partial pressure, indicating that the dissolved concentration of methane is the key kinetic parameter for AOM-D. In order to improve the dissolution of hydrogen, microporous membranes have often been successfully used as a gas diffuser [16, 17]. Similarly, the mass transfer of methane will be one of the key factors to attain denitrification using methane as a sole carbon source.
The overarching goal of this study was to demonstrate the possibility of denitrification using methane as a sole carbon source without an external supply of oxygen, and evaluate its applicability as a post-denitrification technology. Specifically, we investigated 1) the capability of a mixed culture consortium, consisting of anaerobic digested sludge and activated sludge for methanotrophic denitrification under anaerobic condition; 2) kinetic improvement of the microporous membrane especially for the mass transfer of methane; and 3) the performance of bio-reactor coupled with membrane diffuser for methanotrophic denitrification under a non-aerated condition. We also investigated the microbial community structure of the mixed culture in the bioreactor, using fatty acid methyl ester (FAME) methods.
2. Materials and Methods
2.1. Mixed Culture Consortium and Medium
The mixed culture used as a seed for all experiments consisted of activated sludge and anaerobic digested sludge, taken from the ‘J’ wastewater treatment plant (Seoul, Korea) in a 1:1 (w/w) mixing ratio. The growth medium was prepared with the following composition: MgSO4 7H2O, 1,000 mg/L; CaCl2 2H2O, 270 mg/L; FeSO4 7H2O, 9.1 mg/L; 2 mL of phosphate buffer; and 1 mL of trace mineral solution. The composition of buffer and mineral solution in the medium were in the following concentrations: KH2PO4, 24,400 mg/L; Na2HPO4, 10,200 mg/L; FeSO4 7H2O, 2,486 mg/L; MnCl2 4H2O, 500 mg/L; ZnCl2, 50 mg/L; NiSO4 6H2O, 101 mg/L; CoCl2 6H2O, 50 mg/L; Na2MoO4 2H2O, 26 mg/L; H3BO3, 50 mg/L; CuSO4 5H2O, 310 mg/L; and 35% HCl 5 mL [18]. Nitrate was added to the medium solution at 30 and 20 mg NO3−-N/L for anaerobic batch test and bioreactor operation, respectively.
2.2. Batch Test
A 500-mL glass bottle with a rubber screw cap was prepared for the batch test. The working volume was set at 300 mL after adding medium and seed. The initial concentration of the seed was 1,000 mg VSS/L. The batch experiment was carried out under anaerobic condition. In order to remove dissolved oxygen (DO) in the medium, the glass bottle was purged with helium gas for 1 min, and then 20 mL of 99.99% CH4 was added to 200 mL of headspace. The vials were horizontally placed on a rotary shaker and continuously mixed at 400 rpm. The batch experiment was conducted at room temperature (21°C ± 1°C). CH4 and N2 concentrations in the headspace were periodically measured during the experimental period. Liquid sample was taken at the end of experiment in order to measure the NO3−-N concentration.
2.3. CH4 Dissolution Test: Kinetic Assessment
A cylindrical acrylic reactor (10 cm I.D. × 60 cm long, 2.5 L working volume) filled with deionized water was prepared to estimate kinetics for the CH4 dissolution of a microporous membrane diffuser. The dissolution test was carried out by supplying CH4 at various flow rates from 5 to 40 mL/min. The microporous membrane module was designed with four hollow-fiber membranes, which were made of polytetrafluoroethylene (PTFE) with mean pore size of 0.25 μm. The overall surface area of the membrane was 105.73 cm2. A commercial tubular diffuser (Alita industries, Baldwin Park, CA, USA) was simultaneously tested as a control. Compared to the microporous membrane, the poly-ethylene (PE) tubular diffuser had a larger mean pore size and smaller surface area, which were 20 μm and 51.24 cm2, respectively. Liquid sample was collected every 30 min and the concentration of methane was analysed using the headspace method. The volumetric mass transfer coefficient (kLa) of methane was calculated by the following equation:
where kLa is the volumetric mass transfer coefficient (hr−1), C* is the saturated concentration of methane (mg/L), and C is the concentration of methane at each sampling time (t) (mg/L).
2.4. Continuous Operation of Bioreactor Coupled with Membrane Diffuser
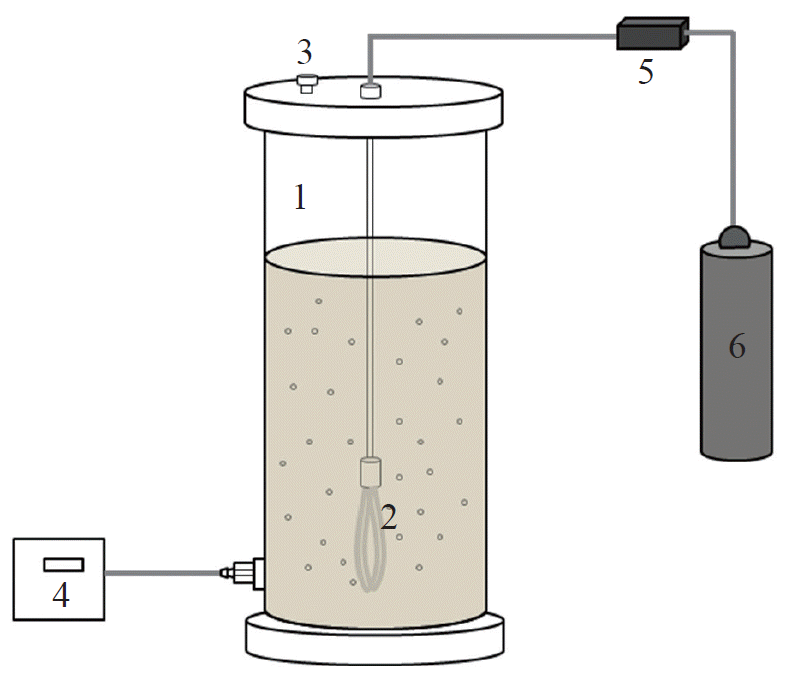
The same reactor (2.5 L) used for kinetic experiment was prepared as a sequencing batch reactor type bioreactor for methanotrophic denitrification under a non-aerated condition (Fig. 1). The bioreactor was operated with a 24-hr fill-and-draw cycle that consisted of a 0.5-hr feeding, 23-hr reaction, and 0.5-hr settling and supernatant decanting period.
A PTFE hollow-fiber membrane diffuser was installed for diffusing methane into the bioreactor. The membrane diffuser was the same one previously tested in the kinetic experiment. Methane was continuously sparged through the membrane at 10 mL/min and the flow rate was controlled by an electric gas flow meter (TSC-210; New Flow System, Seoul, Korea). The reactor was operated under room temperature condition and it was continuously stirred at 400 rpm by magnetic stirrer during the reaction period. The mixed culture used for the batch experiment was used as a seed at 1,000 mg VSS/L at the start of operation. NO3−-N, NO2−-N, and total organic carbon (TOC) in the effluent were periodically measured.
2.5. Analytical Methods
The concentration of CH4 and N2 was analysed using gas chromatography (DS6200; Donam Instruments Inc., Seongnam, Korea) equipped with a thermal conductivity detector using an 8-ft HayeSep Q column (Alltech, USA). The temperatures of oven and detector were 40°C and 120°C, respectively. Helium was used as a carrier gas. NO3−-N, NO2−-N were measured using ion chromatography (ICS-900; Dionex, Sunnyvale, CA, USA) and TOC in the effluent was measured using a TOC analyzer (TOC-V CPH; Shimadzu, Kyoto, Japan).
2.6. Fatty Acid Methyl Esters of Mixed Culture
FAMEs technology was used for analysis of microbial composition in the mixed culture during operation of the bioreactor. The mixed culture sample was collected from the reactor during the reaction period, and then centrifuged at 2,000 rpm for 7 min. Pelleted materials after centrifugation were transferred in 30 mL of glass tube. The 15 mL of 0.2 M methanolic potassium hydroxide was added and pellet was resuspended using a vortex mixer. The glass tube was placed in a 37°C water bath for 1 hr and remixed every 10 min during this period. Approximately 3 mL of acetic acid was added for neutralization. Thereafter, 10 mL of hexane was added to the solution, which was in turn mixed by vortex mixer, and centrifuged at 2,000 rpm for 3 min. The 5 mL of the overlying hexane layer was transferred to a test tube and evaporated to dryness under flowing N2. Finally, the remaining material in the test tube was re-dissolved by 0.5 mL of 1:1 of hexane and methyl tertburyl ether and transferred to a gas chromatography vial. FAMEs were analyzed by gas chromatography (Agilent/HP 6890 series II; Agilent, Santa Clara, CA, USA) equipped with Hewlett-Packard methyl phenyl silicone fused silica capillary column (25 m × 0.22 mm × 0.33 m). The initial oven temperature was programmed from 170°C to 270°C at 5°C/min. Hydrogen gas was used as a carrier gas. The FAME peaks were identified by Sherlock MIS software (MIDI Inc., Newark, DE, USA).
3. Results and Discussion
3.1. Batch Experiment
Batch experiments under anaerobic condition were carried out for 27 days to check the capability of the mixed culture seed for AOM-D, utilizing CH4 as the sole carbon source for denitrification. Fig. 2 shows the variation of CH4 and N2 amount in the headspace during incubation. The CH4 in the headspace was unchanged in 5 days, and then began to decrease. A substantial amount of CH4 appeared to decrease between 5 and 11 days, which accounted for a 57% decrease in CH4 amount. Consequently, CH4 completely disappeared from the headspace at the end of experiment. The maximum CH4 consumption rate was calculated at 0.09 mmol/day. The change in N2 content in the headspace was also identified as shown in Fig. 2. The amount of N2 in anaerobic batch started to increase after 11 days of incubation and reached 1.08 mmol at the end of experiment.
Concentrations of NO3−-N in the liquor were measured for the samples taken at the end of the batch experiment. The amount of NO3−-N remaining was 0.23 mmol, which corresponded to 69% of NO3−-N removal efficiency. The experimentally estimated CH4 utilized for NO3−-N reduction was 1.63, which was about 2.6 times that of the theoretical value (Table 1). The mass balance for nitrogen was calculated by comparing the mass production (0.45 mmol as N) of N2 with the mass depletion of NO3−-N (0.51 mmol). The mass recovery of N2 was 88% indicating that most NO3−-N removed was converted to N2 gas through denitrification. These overall results indicated that the mixed culture used in this study could utilize CH4 as a sole carbon source for denitrification under anaerobic condition, even though the CH4 consumption rate was most likely lower than that of AeOM-D [19].
3.2. Kinetics for CH4 Dissolution of Microporous Membrane Diffuser
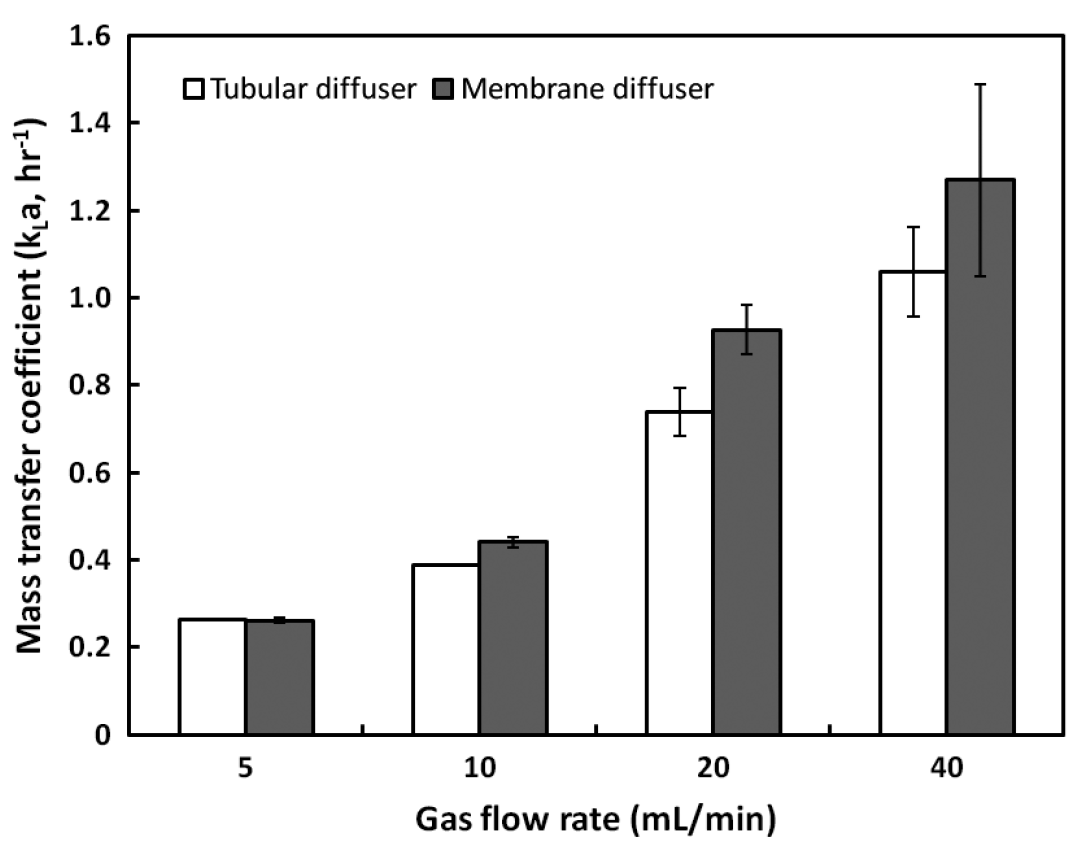
The volumetric mass transfer coefficient (kLa) of methane, depending on flow rates, is shown in Fig. 3. The kLa value describes the rate of dissolution of methane into water and could be affected by the gas component and its concentration, characteristics of the diffuser, and various environmental conditions [20]. As the gas flow rate increased, the kLa steadily increased regardless of the diffuser type and the dissolved methane concentration consequently reached 20.4 ± 0.16 mg/L, which is close to the saturation value (23 mg/L at 20°C). The membrane diffuser showed a higher kLa than the tubular diffuser in all ranges of gas flow rate tested. This was probably attributed to the higher surface area and small pore size of the membrane diffuser than of the tubular diffuser. The smaller pore size could generate finer bubbles, which were most likely well dissolved in the water [21, 22]. The maximum kLa for the membrane and tubular diffuser was 1.27 and 1.06 hr−1 at 40 mL/min, respectively.
3.3. Operation of Non-aerated Bioreactor
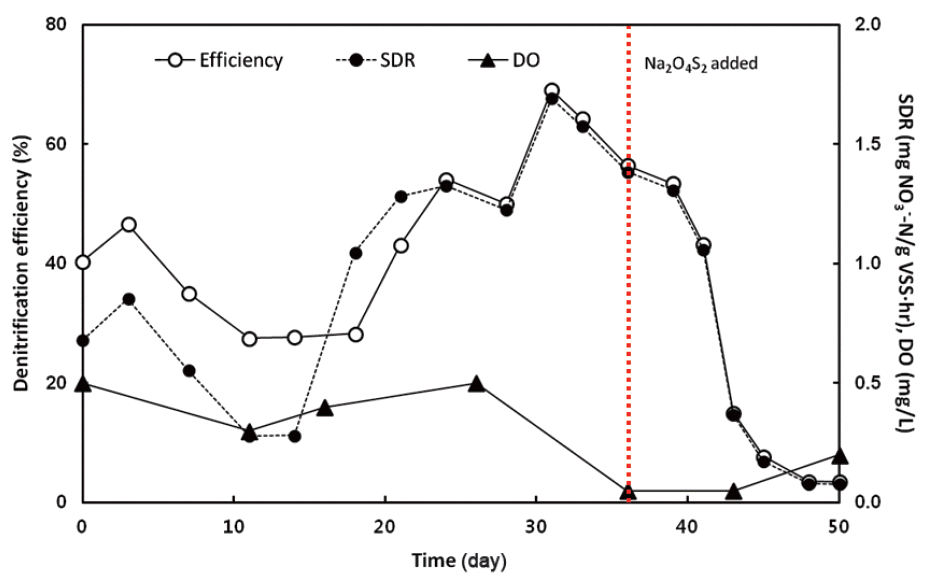
Fig. 4 shows the denitrification efficiency and specific denitrification rate (SDR) obtained from operation of the non-aerated bioreactor for 50 days. The bioreactor was operated without external oxygen (or air) supply, but DO concentration was maintained in the range between 0.3 and 0.5 mg/L due to oxygen remaining in the medium solution. In spite of continuous purging N2 into the medium storage tank, the average DO concentration in the medium tank was 1.0 mg/L.
The denitrification efficiency increased and reached a maximum of 69% after 30 days of operation. In order to check the capability of AOM-D under strict anaerobic condition, sodium dithionite (Na2O4S2) was added at 2 mmol/day after 35 days, and then the DO concentration was rapidly decreased to 0.05 mg/L. Anaerobic condition in the reactor was double-checked by adding resazurin to the effluent as an indicator. The denitrification efficiency drastically dropped as soon as Na2O4S2 was added. The above results indicate that the presence of DO (at least 0.5 mg/L) is necessary to perform denitrification using methane as a sole carbon source, since the direct utilization of methane for AOM-D under strict anaerobic condition is too slow to occur in 23 hr of reaction time. It is generally accepted that low DO is a favourable condition for denitrification, but not for methanotrophic denitrification. Methanotrophic denitrification requires DO for methanotrophs to assimilate carbon via the RuMP or serine pathway [8]. Even though the potential of AOM-D was already confirmed in the anaerobic batch experiment, the rate of methanotrophic reaction was very slow without oxygen [23, 24]. Based on this study and a previous study [25], DO concentration in the bioreactor for methanotrophic denitrification is preferably at a narrow range between 0.5 and 0.8 mg/L, which is achievable in a non-aerated manner. Thus, secondary wastewater effluent would be a good candidate for non-aerated methanotrophic denitrification being applied with inherent DO in the range between 0.9 and 3.7 mg/L [26].
Table 2 shows a comparison of the SDR obtained from the bioreactor with other previous studies. The SDR was very variable among studies, and the maximum SDR of 1.69 mg NO3−-N/g VSS hr obtained from this study was slightly lower than from the previous studies. This was attributable to the following reasons: the high biomass concentration of the mixed culture not pure culture used in this study. However, the obtained SDR showed a similar level to that of another continuous process under aerobic condition [25].
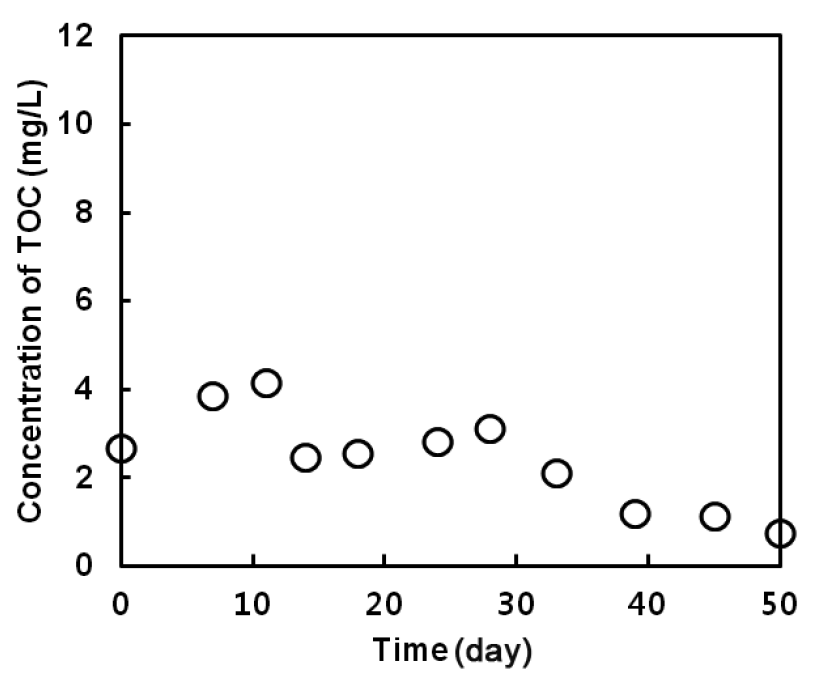
Fig. 5 shows the variation of TOC concentrations in the effluent. One of the most prevalent concerns in the operation of post denitrification is the deterioration of effluent quality due to excessive presence of unutilized organic substances. In methanotrophic denitrification, the remaining organic substances could mainly be dissolved CH4 or methanol. In this study, the TOC concentration averaged 2.45 mg/L during the overall period of operation. In general, the TOC concentration of secondary effluent from most wastewater treatment plants is around 11 mg/L [28]. This result means that concern about residual organic matter after denitrification was not significant in methanotrophic denitrification, since methane or unutilized methanol was not predominant.
This study suggests that methanotrophic denitrification under non-aerated condition can be a promising option for water reclamation of secondary effluent or groundwater remediation with a relatively high denitrification performance and less residual organic concentration. Although the capability of AOM-D under anaerobic condition was not available due to the slow rate, methanotrophic denitrification could be achieved in a bio-reactor without excess supply of oxygen by utilizing the inherent DO in water.
3.4. FAMEs Analysis for Methanotrophs in Bioreactor
Table 3 shows the proportion of major FAMEs extracted from the mixed culture in the bioreactor. Of four major FAMEs identified, the most predominant FAME was C16:1ω7c, which is known to be a typical FAME for type I methanotrophs, such as Methylococcus luteus (UQM3304), Methylococcus whittenburyi (UQM3310), and Methylomonas gracilis [29]. Secondly, the dominant FAME in the mixed culture was C18:1ω7c, which is normally found in an anaerobic digester in high proportion [30]. Also, in several studies, type II methanotrophs have C18 FAME including C18:1ω7c as their major component [29, 31, 32]. Type I methanotrophs are known to be a major microbial group found in most AeOM-D processes; whereas, type II methanotrophs are dominantly found under anoxic or anaerobic condition, with lower DO concentration, compared to methane concentration [33, 34]. This study revealed that both methanotrophs of types I and II were copresent in the bioreactor due to the seed selected (mixed culture of activated sludge and anaerobic digested sludge) and environment (low DO concentration). However, those FAME profiles are also found in other microorganisms as well as methanotrophic bacteria. Hence, in order to more accurately identify methanotrophs in mixed culture, some other methods, based on genomic technology, may be necessary in the future.
4. Conclusions
This study presents the applicability of methanotrophic denitrification under non-aerated condition for the reclamation of secondary wastewater effluent. In batch experiment, methane can be utilized as a sole carbon source for denitrification under anaerobic condition, but rates for methane consumption and nitrate reduction were slow. A microporous membrane diffuser could enhance the mass transfer of methane, so that it was incorporated into the bioreactor. A relatively good denitrification performance was achieved in the bioreactor under non-aerated condition. In spite of no external supply of oxygen, oxidation of methane by methanotrophs occurred by utilizing the residual DO in the influent. The residual TOC concentration in effluents from the bioreactor was much lower than in common secondary effluent. The copresence of two different types of methanotrophs of types I and II in the bioreactor well represented the characteristics of the non-aerated bioreactor providing a favourable circumstance for both types of methanotrophs. The DO concentration in the secondary effluent might be inherently in the range to support methanotrophic denitrification, so that methanotrophic denitrification will be attainable without external oxygen supply. This study suggests that methanotrophic denitrification under non-aerated condition will be a promising option to current post denitrification for the reclamation of secondary wastewater effluent or remediation of groundwater.