AbstractThis study was conducted June 2011 at Gamo Lagoon, after tsunami of March 2011, to estimate food sources and utilization. The results show that the tsunami affected the sediment properties by changing the physical environmental alterations. The fatty acids of the gut content of Nuttallia olivacea mostly comprised the same organic matter found in the sediment. Fatty acids in the tissues showed mainly diatoms, bacteria, and dinoflagellates. That is, most of the food sources (i.e., diatoms, bacteria, dinoflagellates, macroalgae, and terrestrial organic matter) probably pass through the digestive system unharmed; however, terrestrial organic matter, which is refractory to biochemical degradation, indicated a different assimilation trend between the gut content and the tissue. This result suggests that input of labile organic matter from the sediment may control selective metabolism in N. olivacea. From these results, although the physical environment of sediment characteristics by tsunami changed, the food utilization of N. olivacea suggested a better assimilation of selected components from the gut content, irrespective of physical alteration.
1. IntroductionBivalves are not only secondary producers for fish and birds but are also useful bioindicators of environmental changes. In addition, bivalves have water purification abilities, such as retention and removal of particulate organic matter in the water column. This may be particularly important in nutrient and carbon cycling in shallow estuarine areas [1–3]. Some studies have reported on the life history and physiological aspects of Nuttallia olivacea[2, 4, 5]. The infaunal surface-deposit feeder N. olivacea is broadly distributed from Japan, where the water freezes in the winter, to China, which remains warm [6]. This species grows up to 50 mm in shell length and shows a wide range of body sizes [5]. N. olivacea extends a siphon to take in the water column and sediments containing organic materials [6, 7]. In contrast, reports on the ecological characteristics (e.g., food behavior, food utilization) of N. olivacea are rare.
Surface-deposit feeders, such as N. olivacea, may use various organic matters from the water column and sediment, since the particulate organic matter in estuarine tidal flat contains various matters. Previous studies have shown that microbenthic algae and marine phytoplankton are important food sources for bivalves in estuarine ecosystems, because the former are highly productive and nutritious [8–10]. Especially, the food sources of N. olivacea reported mainly benthic diatoms, bacteria, and suspended solids in the Nanakita estuary, Japan [7, 9]. These results suggest that the feeding activity of N. olivacea depends on the habitat. Therefore, the physical environmental alterations (e.g., tsunami and earthquake) can strongly influence the food utilization of N. olivacea. However, the relationship between the food availability of N. olivacea and sediment properties remains unclear.
A tsunami is a rare disturbance within marine communities [11], but it has a high intensity and/or energy [12, 13]. In addition, tsunami waves can resuspend, transport, and redeposit sediments on the sea floor, as a result of repeated incoming and outgoing flows [14]. These processes have great potential to affect coastal benthic communities and the physical environment. In particular, the biological environment is directly or indirectly modified by tsunami in that alters the local abundance of refuges, food, or nutrients available to resident species or others that interact with them [11]. On March 11, 2011, an earthquake with a moment magnitude (Mw) of 9.0 occurred off the coast of Japan (142°51′ E, 38°06′ N) at a depth of 24 km. The Gamo Lagoon (Fig. 1) is an area located in Sendai Bay. Thus this study area was influenced by tsunami waves. As a result, flooding occurred along a stretch of coastline from Sendai Bay to the city of Sendai.
After observing this event we hypothesized that a tsunami had the potential to negatively affect the benthic environment and would result in changes to food sources for macrobenthos (i.e., N. olivacea). Thus, the main objective of this study was to evaluate the immediate effect of the tsunami on food sources of N. olivacea located at Gamo Lagoon of the Nanakita River estuary, Japan. The utilization of the food sources for N. olivacea is discussed in relation to the change of sediment environment in a brackish Gamo Lagoon on the basis of the fatty acid analysis.
2. Materials and MethodsThis study was conducted in a tidal flat system of the brackish Gamo Lagoon (0.11 km2) of the Nanakita River estuary on the northeastern coast of Honshu Island, Japan (Fig. 1). The minimum water depth at station B was 10 cm at mean low water. Three sampling stations, A–C, were established in the intertidal zones of the tidal flat system. All samples were collected on June 6, 2011. Surface sediments were collected using acrylic core samples (10 cm in diameter and 20 cm in depth). The sediment at 0–1 cm depth was analyzed in the laboratory, for fatty acids, chlorophyll α (Chl α), carbon-to-nitrogen (C/N) ratio, silt-clay content, and total organic carbon (TOC) content. All sediment organic matter (SOM) samples were immediately freeze-dried in the laboratory and then ground into powder using a mortar and pestle prior to analysis. Sampling of the N. olivacea was conducted at station A–C (i.e., nine points, Fig. 1). Three sediment cores from the surface to a depth of 20 cm were collected using an acrylic core at each station. N. olivacea samples were washed with distilled water to remove extraneous matter from the tissues prior to analysis. Then, all of the body tissues of N. olivacea were used for the fatty acid analyses. The gut sections of N. olivacea in each station were placed in clean glass Petri dishes in a refrigerator until fully thawed, after which the gut content was sucked up with a sterile pipette and transferred to sterile vials. Maximum care was given to avoid extracting any gut tissue during dissection and removal of the gut content. Separate gut contents were used for the fatty acid analyses. The N. olivacea is a dominant species and shows wide distribution within different environments in Nanakita estuary and Gamo Lagoon [6]. The mean densities (531 ind/m2) of N. olivacea in the Gamo lagoon were high compared to Ruditapes philippinarum (105 ind/m2) and Macoma contabulata (183 ind/m2) [9]. This is a good tool to discuss its role in the biogeochemical cycle in the study area.
Tissue and gut contents of N. olivacea, SOM were used following the method of Abdulkadir and Tsuchiya [15] for lipid extraction. The samples were mixed with 8 mL of hexane and then 1 mL of internal standard solution in a 50 mL centrifuge tube. The centrifuge tube was flushed with nitrogen gas and then closed tightly with a screw cap after 2 mL of BF3 in methanol was added and stirred using a mixer. The centrifuge tube was heated in a water bath at 100°C for 120 min. After being cooled to room temperature, 1 mL of hexane was added, followed by 2 mL of distilled water. The centrifuge tube was then shaken vigorously for 1 min using a mixer and centrifuged for 3 min at 2,500 rpm. The upper phase of the two phases that formed was the hexane layer containing fatty acid methyl esters (FAMEs). Finally, 1 mL of the hexane layer was transferred using a Pasteur pipette into a clean sample vial to be injected into the gas chromatograph (GC) for FAME analysis. The FAMEs were separated and quantified using a GC (GC-2014; Shimadzu, Kyoto, Japan) equipped with a flame ionization detector. A free fatty acid phase (FFAP)-polar capillary column (100 m × 0.25 mm internal diameter) was used for the separation with a helium carrier gas. After injection at 150°C, the oven temperature was raised to 150°C at a rate of 1°C per min, then to 220°C at 5°C per min, and was finally held constant for 50 min. The flame ionization was held at 250°C. Most FAME peaks were identified by comparing their retention times with those of authentic standards (Supelco Inc., Bellefonte, PA, USA). Fatty acid peaks were identified using GC-mass spectrometry for some samples. Our assessment focused primarily on fatty acid markers representing five major organic sources for SOM: diatoms, bacteria, green algae, dinoflagellates, and fatty acids with more than 24 carbons (long chain fatty acids [LCFAs]) from terrestrial plants (Table 1) [16, 17]. The fatty acid biomarkers of these major organic sources were identified in SOM by comparison to a standard (Supelco 37 component FAME). The relative abundances of the major organic sources were calculated (percent of total).
Samples for TOC and C/N ratio analysis were dried (105°C, 24 hr), homogenized, and then treated with 10% HCl to remove carbonate. The TOC and C/N ratios were determined using a Heraeus vario-EL CHN-analyzer (vario-EL III; Elementar Analysensysteme GmbH, Hanau, Germany). Chl α in the SOM samples was extracted using 90% acetone, and their concentrations were determined by spectrophotometric measurement according to the method of Lorenzer and Jeffrey [18]. Sediment types were determined, according to a classification scheme based on sediment mud content (i.e., the percentage of the sediment fraction <63 μm to total sediment dry weight) [19].
TOC content, Chl α, total fatty acid, and major organic matter fatty acids in sediment collected from different stations were compared using one-way analysis of variance (ANOVA) with spatial sampling occasions as factors. Correlation analyses were conducted to examine the relationships among major organic sources of SOM, gut content, and tissue. The Statistical Package for the Social Sciences ver. 17.0 (SPSS Inc., Chicago, IL, USA) was used for the statistical analyses.
3. Results and Discussion3.1. Changes of Sediment Organic Matter by TsunamiThe TOC (0.05%–0.38%) and silt-clay (0.04%–1.97%) results indicated a changing trend from station A toward station C in Gamo Lagoon (Table 2). Sediments of the Gamo Lagoon (Stations A, B, and C) were characterized as pure sand with low mud content (sediment fraction <63 μm was less than 2%) according to the sediment classification of Flemming and Delafontaine [19]. The percentage of silt-clay and TOC content (Station B) after tsunami was lower than before (station b). Meanwhile, the C/N ratios of the surface sediment were not significantly different between stations A, B, and C (Table 2). These C/N ratios (6.7–8.2) of SOM in between stations provide an indication of the quality of organic matter. The C/N ratio for marine-produced organic matter was 6.0 ± 0.36 [20]. The main component of SOM in the present study is regarded as microalgae or its detritus, because this value is comparable to that the C/N ratio of microalgae ranges from 3 to 8 in general [21]. These results revealed that the amount and origin of organic matter in the surface sediment. Chl α concentrations in the SOM were 2.98 μg/g (station A), 7.48 μg/g (station B), and 8.40 μg/g (station C), respectively (Table 2). The spatial distributions (about 3–8.4 μg/g) of Chl α indicated that its concentration increased with increase the in sediment mud content (r2 = 0.617, p < 0.1). However, these results indicated different sediment characteristics in the Gamo Lagoon, comparison to the previous study [9]. According to Kanaya et al. [9], the silt-clay, TOC, Chl α, and C/N ratio of sediments indicated 3%–4.5%, 1.2%–2.2%, 3.5–7.5 μg/g, and 8–9.5, respectively. From these results, the surface sediment of Gamo Lagoon before the tsunami had a higher contribution of terrestrial organic matter compared to after the tsunami (June 2011). Conversely fatty acid biomarker analysis is generally able to provide an insight into the specific sources of organic matter in estuarine environments [17, 22]. In the present study, the strong representation of diatoms in the SOM was determined by the high contribution of biomarker 20:5ω3, which is considered to be an indicator of benthic diatoms [23]. The distribution of diatoms was higher from station A toward station C of the Gamo Lagoon in Fig. 2(a). In general, the benthic diatoms biomass in sandy habitats is lower than in muddy habitats due to limited nutrients [24, 25]. Moreover, Fatty acid contents of microalgae (e.g., diatom and dinoflagellates) in the sediment closely correlated with those of Chl α, suggesting that benthic microalgae and marine phytoplankton might be the main contributor of fatty acids in the SOM. Fatty acid biomarkers of bacteria showed a trend similar to that of microalgae (Table 3). A similar trend was observed in Altamaha estuarine sediments [26]. The effect of LCFAs was different at all the stations. This result suggested the importance of terrestrial plants as one of the main sources of organic matter in estuarine tidal flats, with great potential for export to adjacent habitats [27].
Our results show that the soft bottom sediment of the study site has been affected by the disturbance produced by an earthquake and subsequent tsunami. The tsunami event produced changes in sediment granularity, enhancing silt sediments, indicating their recent transport [28].
3.2. Selective Assimilation of Food Sources by N. olivaceaCompared to other fatty acids, fatty acids specific for diatoms and dinoflagellates were more abundant in N. olivacea collected at the study stations in Fig. 2(b) and (c). Although the concentrations of dinoflagellate fatty acids from the gut significantly differed between stations A and C (Fig. 2(b); p > 0.05), analysis in the tissues did not significantly differ between stations. LCFAs, terrestrial plant-derived organic matter, were detected in the gut content (Fig. 2(b)). However, fatty acids of LCFAs (e.g., terrestrial plant-derived organic matter) were not detected in tissues of N. olivacea (except for station C; Table 4). In the present study, we observed that fatty acid biomarker distributions in tissues of N. olivacea collected from different stations in the Gamo Lagoon estuarine area varied in a similar manner to those in gut content and sediments, suggesting that input of labile organic matter from the sediment may control selective metabolism [29]. Thus, fatty acid biomarkers of food sources in the gut content of N. olivacea were highly correlated with sediment (Table 3). N. olivacea obtain potential food particles via filtration, by holding their inhalant opening or siphon above the surface sediment. However, they may not always inhale water. In fact, they live in the sediment and do not hold their inhalant siphon above the sediment when the tidal flats are exposed. Therefore, the digestive tract contained the same microorganisms as the water and local sediments. The stomach and intestinal contents of bivalve specimens investigated by Korinkova [29], mostly contained the same microorganisms as those found in the surrounding environment. The inorganic particles are probably rejected in the form of pseudofeces due to their size and weight. A detailed analysis of the pseudofeces should be performed to confirm this assumption. In N. olivacea tissues, variations in fatty acid bio-markers of diatoms and dinoflagellates were similar to that in the gut content (Table 3). That is, this species shows active selection by preferential ingestion and/or better assimilation of selected components from the gut content. Conversely, the relation of terrestrial plant fatty acid biomarkers in the gut content and tissue differed from the relationship in the sediment and gut content, since terrestrial plants contain lignin and cellulose, which are refractory to biochemical degradation. The present study confirmed the absence of any special separation mechanisms that would allow N. olivacea to preferentially intake utilizable food sources. Microalgae and bacteria of suitable size that are re-suspended in the sediment pass into the N. olivacea gut content. Some of them are then disintegrated by digestive enzyme action without disruption. The results suggest that microalgal-derived organic matter or bacteria are the main source of nutrients for N. olivacea. A similar trend was observed in the previous study of Gamo Lagoon [9]. These results indicate that the microalgae were important as food sources of N. olivacea.
3.3. Utilization Characteristics of Food Source Before and After the Tsunami in N. olivaceaThe food utilization of N. olivacea was more influenced by the depositional environment in estuarine tidal flat according to the investigation by Shin et al. [7]. That is, N. olivacea is considered to select and assimilate organic matter. These results were in agreement with the findings of the present study. From these results, the change of utilization of food sources in N. olivacea was not solely related to the magnitude of the tsunami disturbance. The tsunami event did not produce differences in the diversity and abundance according to some studies [28, 31]. However, other studies demonstrated that the benthic communities are affected by natural (or anthropogenic) conditions (melt down and radioactive release) [32, 33]. These studies revealed that the changes in physical environment of sediment characteristics before and after the tsunami varied between sites. Therefore, more research is needed to more precisely identify the effects of tsunami in the relationship between the macrobenthos and their habitat.
4. ConclusionsWe conclude that the tsunami did not change food utilization in N. olivacea, but we found differences in the amount and origin of the sediment before and after the disturbance. N. olivacea prefer microorganisms (e.g., diatom, dinoflagellates, and bacteria) as food sources to terrestrial organic matter. This may highlight the importance of microalgae for N. olivacea, such as in retarding or increasing levels of physical disturbance in the face of global climate changes and in maintaining the stability of biodiversity and ecosystem function.
AcknowledgmentsWe appreciate the help of H. Nomura, M. Fujibashi, and Y. Nagahama for preparing and analyzing the fatty acid samples. This work was supported by KAKENHI (21360249, 23760497) and the Environmental Research and Technology Development Fund (B-1004) of the Ministry of the Environment, Japan.
References1. Kasai A, Toyohara H, Nakata A, Miura T, Azuma N. Food sources for the bivalve Corbicula japonica in the foremost fishing lakes estimated from stable isotope analysis. Fish. Sci. 2006;72:105–114.
2. Kanaya G, Kikuchi E. Spatial changes in a macrozoobenthic community along environmental gradients in shallow brackish lagoon facing Sendai Bay, Japan. Estuar. Coast. Shelf Sci. 2008;78:674–684.
3. Sakamaki T, Richardson JS. Dietary responses of tidal flat macrobenthos to reduction of benthic microalgae: a test for potential use of allochthonous organic matter. Mar. Ecol. Prog. Ser. 2009;386:107–113.
4. Tomiyama T, Ito K. Regeneration of lost siphon tissues in the tellinacean bivalve Nuttallia olivacea. J. Exp. Mar. Biol. Ecol. 2006;335:104–113.
5. Tomiyama T, Omori M. Interactive effects of sublethal predation and body size on shiphonsiphon production of the bivalve Nuttallia olivacea. J. Exp. Mar. Biol. Ecol. 2007;341:102–109.
6. Tsuchiya M, Kurihara Y. Effect of the feeding behaviour of macrobenthos on changes in environmental conditions of intertidal flats. J. Exp. Mar. Biol. Ecol. 1980;44:85–94.
7. Shin WS, Fujibayashi Y, Nomura M, Nakano K, Nishimura O. Fatty acid composition between Nuttallia olivacea and Hediste spp. in the Nanakita Estuary, Japan: estimation of food sources. J. Water Environ. Technol. 2012;10:11–22.
8. Yokoyama H, Tamaki A, Koyama K, Ishihi Y, Shimoda K, Harada K. Isotopic evidence for phytoplankton as a major food source for macrobenthos on an intertidal sandflat in Ariake Sound, Japan. Mar. Ecol. Prog. Ser. 2005;304:101–116.
9. Kanaya G, Nobata E, Toya T, Kikuchi E. Effects of different feeding habits of three bivalve species on sediment characteristics and benthic diatom abundance. Mar. Ecol. Prog. Ser. 2005;299:67–78.
10. Dubois S, Orvain F, Marin-Leal JC, Ropert M, Lefebvre S. Small-scale spatial variability of food partitioning between cultivated oysters and associated suspension-feeding species, as revealed by stable isotopes. Mar. Ecol. Prog. Ser. 2007;336:151–160.
11. Sousa WP. Natural disturbance and the dynamics of marine benthic communities. Bertness MD, Gaines SD, Hay ME, editorsMaine community ecology. Sunderland: Sinauer Associates; 2001. p. 85–130.
12. Krishankutty N. Effects of 2004 tsunami on marine ecosystems: a perspective from the concept of disturbance. Curr. Sci. 2006;90:772–773.
13. Bourrouilh-Le Jan FC, Beck C, Gorsline DS. Catastrophic events (hurricanes, tsunami and others) and their sedimentary records: introductory notes and new concepts for shallow water deposits. Sediment. Geol. 2007;199:1–11.
14. Noda A, Katayama H, Sagayama T, et al. Evaluation of tsunami impacts on shallow marine sediments: an example from the tsunami caused by the 2003 Tokachi-oki earthquake, northern Japan. Sediment. Geol. 2007;200:314–327.
15. Abdulkadir S, Tsuchiya M. One-step method for quantitative and qualitative analysis of fatty acids in marine animal samples. J. Exp. Mar. Biol. Ecol. 2008;354:1–8.
16. Napolitano GN, Pollero RJ, Gayoso AM, MacDonald BA, Thompson RJ. Fatty acids as trophic markers of phytoplankton blooms in the Bahia Blanca estuary (Buenos Aires, Argentina) and in Trinity Bay (Newfoundland, Canada). Biochem. Syst. Ecol. 1997;25:739–755.
17. Alfaro AC, Thomas F, Sergent L, Duxbury M. Identification of trophic interactions within an estuarine food web (northern New Zealand) using fatty acid biomarkers and stable isotopes. Estuar. Coast. Shelf Sci. 2006;70:271–286.
18. Lorenzer CJ, Jeffrey SW. Determination of chlorophyll in sea-water: report of intercalibration tests. Paris: United Nations Educational, Scientific and Cultural Organization; 1980.
19. Flemming BW, Delafontaine MT. Mass physical properties of muddy intertidal sediments: some applications, misapplications and non-applications. Cont. Shelf Res. 2000;20:1179–1197.
20. Kasai A, Horie H, Sakamoto W. Selection of food sources by Ruditapes philippinarum and Mactra veneriformis (Bivalva: Mollusca) determined from stable isotope analysis. Fish. Sci. 2004;70:11–20.
21. Meyers PA. Preservation of elemental and isotopic source identification of sedimentary organic matter. Chem. Geol. 1994;114:289–302.
22. Shi W, Sun MY, Molina M, Hodson RE. Variability in the distribution of lipid biomarkers and their molecular isotopic composition in Altamaha estuarine sediments: implications for the relative contribution of organic matter from various sources. Org. Geochem. 2001;32:453–467.
23. Parsons TR, Takahashi M, Hargrave B. Biological oceanographic processes. Oxford: Pergamon Press; 1984.
24. Koster M, Meyer-Reil LA. Characterization of carbon and microbial biomass pools in shallow water coastal sediments of the southern Baltic Sea (Nordrugensche Bodden). Mar. Ecol. Prog. Ser. 2001;214:25–41.
25. MacIntyre HL, Geider RJ, Miller DC. Microphytobenthos: the ecological role of the “secret garden” of unvegetated, shallow-water marine habitats. I. Distribution, abundance and primary production. Estuaries. 1996;19:186–201.
26. Dai J, Sun MY. Organic matter sources and their use by bacteria in the sediments of the Altamaha estuary during high and low discharge periods. Org. Geochem. 2007;38:1–15.
27. Woodroffe CD. Studies of a mangrove basin, Tuff Crater, New Zealand. I. Mangrove biomass and production of detritus. Estuar. Coast. Shelf Sci. 1985;20:265–280.
28. Lomovasky BJ, Firstater FN, Salazar AG, Mendo J, Iribarne OO. Macro benthic community assemblage before and after the 2007 tsunami and earthquake at Paracas Bay, Peru. J. Sea Res. 2011;65:205–212.
29. Korinkova T. Food utilization in fingernail and pill clams. Malacol. Bohemoslov. 2011;10:1–4.
30. Shin WS, Aikawa Y, Nishimura O. Chemical properties of sediment in Nanakita estuarine tidal flat: estimeation of sedimentary organic matter origin by stable isotope and fatty acid. Environ. Eng. Res. 2012;17:77–82.
31. Whanpetch N, Nakaoka M, Mukai H, et al. Temporal changes in benthic communities of seagrass beds impacted by a tsunami in the Andaman Sea, Thailand. Estuar. Coast. Shelf Sci. 2010;87:246–252.
Fig. 2Spatial variations of food sources of sediments and Nuttallia olivacea in Gamo Lagoon: (a) sediment, (b) gut content, and (c) tissue. Graphs show the mean with the standard deviation (n = 3). Different letters indicate significant (p < 0.05) differences between those values. *By Shin et al. [7]. 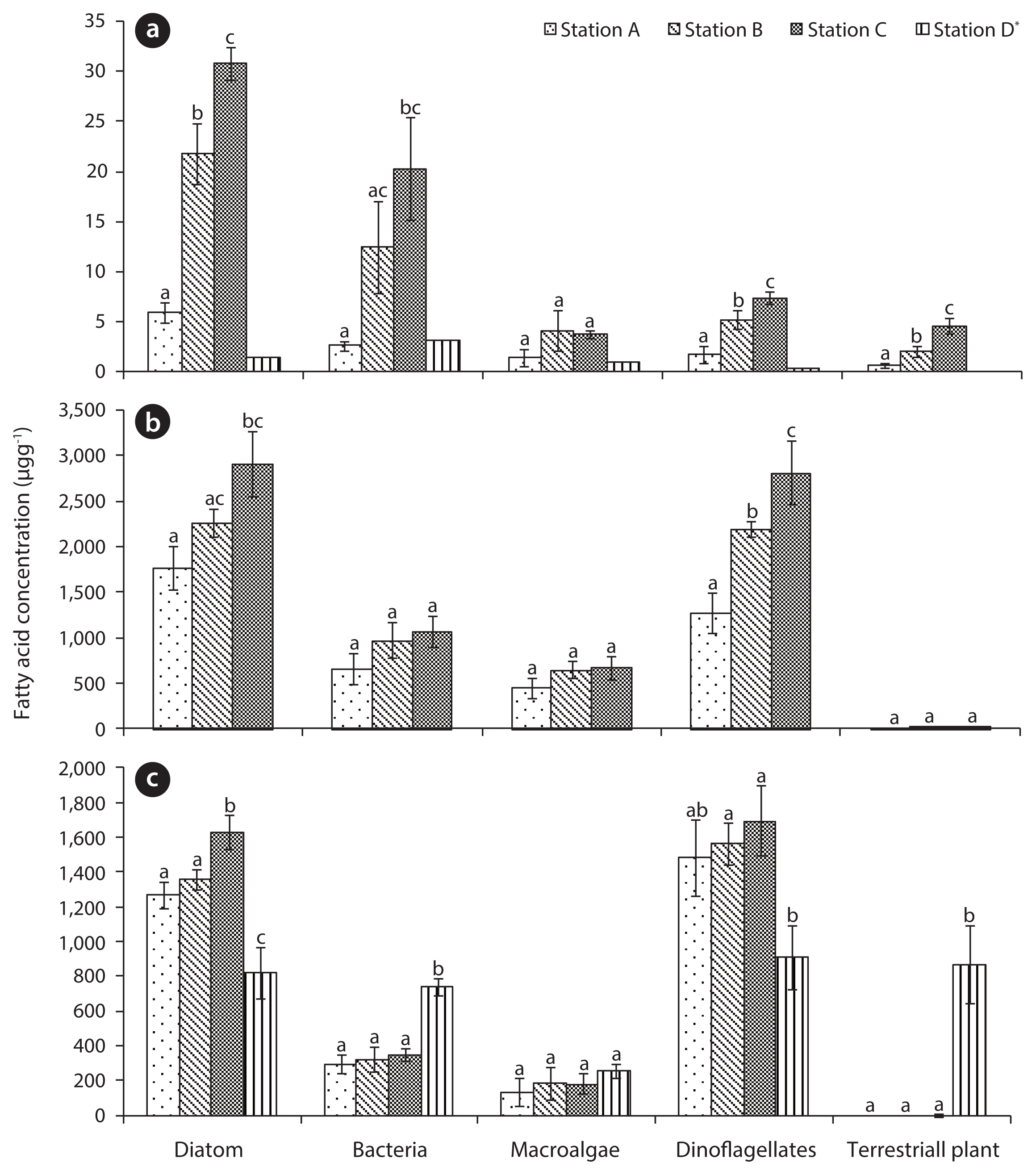
Table 1Fatty acids used as trophic marker for different food sources
Table 2Sediment characteristics of the three stations investigated in the Gamo Lagoon
Table 3Correlation coefficient between each potential organic matter on sediment, gut content, and tissue in Gamo Lagoon
Table 4Spatial change of selected fatty acid (FA) in sediment, gut content and tissue
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||