AbstractThe present study focused on the various advanced oxidation processes; Ozone, UV radiation, O3/H2O2, O3/UV, UV/H2O2 and O3/UV/H2O2 for treatability of dairy industry wastewater. With this aim, the trials were carried out in cylindrical reactor fortified with UV radiation and Ozone injection. Efficiency of the treatment process was evaluated considering Chemical Oxygen Demand (COD), lactose reduction and process parameters were determined to be reaction time, pH, circulation rate, and H2O2 dosage. 32.5%, 35.2%, 25%, 83% COD and 40.6%, 43.6%, 38.2%, 80% lactose reduction efficiency were obtained under the operating conditions for O3/H2O2, O3/UV, UV/H2O2 and O3/UV/H2O2 processes, respectively. As per this outcome, UV/H2O2/O3 process gave more than 65% of COD and 52.36% of lactose reduction efficiency than other hybrid processes. Optimum conditions for UV/H2O2/O3 process (pH = 5, time = 180 mins, circulation rate = 50 mL/h and H2O2 dosage of 0.5 mL) resulted in 88% of COD and 93.4% lactose reduction.
1. IntroductionAmong the food industrial sectors, dairy industry consumes more amount of water. Dairy plants include milk processing, butter manufacturing, cheese/whey manufacturing, ice-cream manufacturing and milk powder productions were generating the maximum volume of effluent in a day by day activities [1]. In India, approximately one liter of milk processing produces 10 L of effluent and it may vary in different countries and dairy plants [2]. Wastewater from these dairy plants composed of carbohydrates, protein, fats, nutrients, sanitizers and detergents [1]. Among these one kg of protein produces 1.36 kg of COD, lactose produces 1.13 kg of COD, fat produces 3 kg of COD [3]. The pollution load was represented in terms of Biological Oxygen Demand (BOD) (40–48,000 mg/L), Chemical oxygen demand (COD) (80–95,000 mg/L), Total Suspended Solids (TSS) (24–88,000 mg/L) and pH (4.2–9.4) and its values could be varying with season conditions [4, 5]. Discharging of dairy effluents to water bodies prior to treatment creates various human diseases such as yellow fever, dengue fever, chicken guniya, diahria, lunk cancer and kidney failure etc., [6]. Continuous degradation of casein, lactose and presence of microorganisms create oxygen depletion in receiving water body causes odor, turbidity and eutrophication which was hazardous to fish and algae [5, 7]. The selection and design of proper treatment methods were essential to increase the treatment efficiency of dairy effluent plants [8].
Biological processes such as aerobic and anaerobic, physicochemical processes such as electrocoagulation, membrane process and adsorption were widely used to treat the dairy effluent due to the presence of rich in organic matter [9, 10]. However, the above-mentioned treatment processes produce maximum amount of sludge, periodical maintenance, hydraulic/solid retention time, requirement of pretreatment, cost of electrodes/coagulants/adsorbents, recovery of coagulants/adsorbents, difficulties faced in disposal of sludge, cost of electricity and membrane, periodical replacement of electrode/membrane and toxicity of effluent leads to limitation of the process [11–14]. Although these methods produce considerable results in pollutant removal, its not give complete elimination of pollution load in dairy effluents.
In order to achieve the complete degradation of pollutants, the usage of Advanced Oxidation Methods (AOPs) in dairy wastewater was considerably increasing. In AOP, the generation of highly active chemical species as hydroxyl radicals which has a tendency to react with pollutants and converted into CO2 and H2O [15, 16]. Ozone was one of the AOP, which was directly or indirectly (in the form of secondary radicals) reacts with organic compounds [17]. As the ozone alone in dairy treatment process should not promote complete mineralization of organic compounds. Hence, the efficiency of ozone process was enhanced by the addition of catalytic reactants such as UV radiation, fenton and H2O2 [18–20]. Nowadays most of the researchers were concentrating on AOP due to the following advantages such as generation of ozone consumes less energy, no sludge production, decrease in volume of sample, less treatment time, gaseous state, easy maintenance, simple and reliable operation which considerably increased the treatment performance of dairy wastewater [18, 19, 21]. In the AOP process, compared to an individual, hybrid processes such as O3/H2O2, O3/UV and UV/H2O2 processes were mostly used to treat the different industrial wastewater includes hospital, pharmaceutical, winery, olive oil and textile [22]. Although AOP has notable merits, the presence of residual ozone and un-oxidized ozone from the reactor could create harmful effects on the human being. Proper installation of ozone destruction system in wastewater treatment plants considerably reduces the above-mentioned problems [23].
Efficiency of combined AOP process depends upon the buffering conditions of effluent, concentration of pollutants, temperature, Total Solids (TS), TSS, turbidity, COD and BOD load, ozone concentration, ozone diffusion rate and ozone decomposition rate [24, 25]. The combination of AOP with Biological or physicochemical methods superiorly promotes the treatment performance which creates a possible way to achieving zero pollutant discharge. László, Kertész [26], also proved that the pre ozone treatment with nanofiltration greatly increases the biodegradability of dairy wastewater to 96.4%. This was due to the application of preozonation considerably reduces the membrane flux that could promote the filtrate properties of membrane [27]. In contrast, when applying a fenton as a post-treatment of membrane process, it does not give possible results in pollutant removal due to the formation of ion compounds during fenton reaction [28, 29].
This present study aims to investigate the performance of synergisation of UV radiation with ozone/H2O2 system in simulated and real dairy effluent. The effect of various parameters such as circulation rate, H2O2 concentration and pH on pollutant removal interms of BOD, COD, turbidity and color removal were studied. The real dairy industry effluent was treated under the optimum conditions obtained in simulated dairy effluent treatment.
2. Materials and Methods2.1. Dairy Wastewater and its CharacterizationReal time dairy industrial wastewater was collected from the local industry located in Tamilnadu, India and the samples were collected in accordance with standard methods of sampling. Polyethene cans were properly washed with mild detergent and then leached with 1:1 HCl overnight. The containers were rinsed with deionized water before the sampling at the point of discharge. Two different sets of samples were taken in 1,000 mL bottles initially and then poured into other two different five liter cans for homogenization. All the samples were stored in ice box containing well frozen ice until they were taken to the laboratory.
Similarly, simulated dairy wastewater was prepared by dissolving 4 g of skimmed milk powder in one liter of distilled water [30–32]. The milk powder used were of commercial grade and used throughout the experiments without further purification in order to represent the actual conditions. Per 100 g of skimmed milk powder consists 22 g of protein, 18 g of fat and 50 g of carbohydrates. To achieve uniformity throughout the experiment, simulated dairy wastewater was newly prepared before the experimental studies. The prepared simulated dairy wastewater was considered as a representative sample equivalent to the characteristics of real time dairy effluent.
2.2. Experimental SetupThe experimental setup for Ozone/UV/H2O2 system was shown in Fig. 1. It was divided into five parts: (1) Photo reactor, (2) H2O2 feed tank, (3) Circulation tank, (4) Ozonator, and (5) Ozone destructor. The photoreactor was made up of glass tube with a diameter of 3 cm and a length of 20 cm. The photoreactor was equipped with the provision for the insertion of UV lamp (Philips) of 16 W power supply 254 nm wavelength at the top of the reactor and ozone gas from the ozonator (Faraday ozone FVP10–136) was introduced from bottom of the reactor through a diffuser at the rate of 800 mg/h. Ozone destructor was used to destruct the unreacted ozone coming out from the reactor. The required quantity of H2O2 was fed into the reaction chamber from the peroxide tank during circulation of the simulated dairy effluent. A programmable peristaltic pump was used to circulate the dairy wastewater at a different flow rate into the reactor at room temperature. A simulated dairy wastewater sample was drawn at a definite time interval from the reaction chamber to analyze the performance of the system during the course of the treatment process.
In the present study, the treatment efficiency of system was studied by varying operational parameters such as pH from 5 to 9, H2O2 concentration from 0.5 to 0.15 mL/L, circulation rate from 50 to 150 mL/h at a fixed ozone dosage of 800 mg/h. pH of the simulated dairy effluent was adjusted using sodium hydroxide and hydrochloric acid. All the experimental trials were carried out in a circulation mode with a sample volume of 160 mL.
2.3. Analytical Methods and DeterminationThe measurable parameters such as pH, conductivity, Chemical Oxygen Demand (COD), Biological Oxygen Demand (BOD), turbidity, Total Dissolved Solids (TDS), turbidity and lactose were measured in dairy wastewater before and after the treatment to ensure removal efficiency of the system. Standard analytical methods were used as per IS 3370 and APHA standards. The pH and conductivity were measured by using digital pH meter (Elico LI 120) and conductivity meter (Elico CM 180). Potassium dichromate and sulfuric acid were used to determine the COD. TDS was determined using TDS analyzer (Elico CM 183). Lactose content present in the sample was analyzed spectrophotometrically using Elico CL 223 Colorimeter. Nephelometer (Elico CL 52D) was used to determine the turbidity of the sample. DO meter (Hach HQ430d flexi) was used to analyze BOD present in the dairy effluent.
3. Results and Discussion3.1. O3/H2O2, O3/UV and UV/H2O2 SystemThe preliminary experimental work was carried out by using advanced oxidation processes such as ozone, UV and H2O2 either in individual or combination of these processes. From Fig. 2 (a) and (b), when adopting ozone, UV and H2O2 process individually, COD and lactose reduction efficiency was less than l0% which was in agreement with the literature Slavov [34]. However, the combined hybrid process such as O3/H2O2, O3/UV and UV/H2O2 was carried out at a pH level of 7 and these processes gave notable COD and lactose reduction in dairy wastewater treatment was shown in Fig. 2 (a) and (b).
In O3/H2O2 system, addition of H2O2 in wastewater considerably enhances the COD and lactose reduction of 32.5% and 40.61%, respectively. When H2O2 combined with ozone, the ozone was decomposed into OH* radicals and form a peroxone process in aqueous solution. The H2O2 was a strong oxidizer having an oxidative potential of 1.78 eV and fastly react with HO2− ions rather than O3 and generates more OH* radicals as seen in the following reaction [22] (Eq. (1)–(3)).
It can be seen that the O3 disintegrates into two OH* radicals which generate more OH* radicals than ozone alone process with the presence of H2O2 oxidant. The efficiency of COD and lactose reduction was depended upon the H2O2 dosage.
In O3/UV system, COD of 35.2% and lactose of 43.68% reduction was attained during the 3 h treatment process. When ozone was directly irradiated by UV, a maximum quantity of OH* radicals were produced than UV or Ozone processes in individuals. As from the following reaction, the photodegradation of O3 molecules in water produces H2O2 and O2 ions further decomposed into OH* radicals [22] (Eq. (4)–(5)).
UV/H2O2 system also gave a considerable result in COD and lactose reduction of 25% and 38.25%, respectively (Fig. 2 (a) and (b)) which was slightly higher than the O3/H2O2 and O3/UV system. When H2O2 was exposed through UV radiation in solution phase, the OH* radicals were generated in accordance with the following reaction [22] Eq. (6).
As of hybrid processes such as O3/H2O2, O3/UV and UV/H2O2, the O3/UV process gave slightly better results in COD and lactose reduction than other two processes of O3/H2O2 and UV/H2O2 was proved by Pourgholi, Jahandizi [35].
3.2. UV/H2O2/O3 SystemDuring the process, decomposition of H2O2 produces more secondary species such as exited superoxides, hydrogen peroxide and hydrogen atoms. Although this chemically formed secondary species have a tendency to react with organic matter in the dairy wastewater. The combined use of UV/H2O2/O3 in dairy wastewater enhances the OH* radicals formation which ultimately increases the COD and lactose reduction. In this present study, UV/H2O2/O3 system was further analyzed at the different operational parameters (circulation rate, reaction time, initial pH and H2O2 dosage) and verified with COD and lactose reduction.
It can be explained by the fact that addition of UV and H2O2 into O3 system greatly enhances degradation efficiency. UV irradiation accelerates the decomposition rate of O3 and H2O2 into hydroxyl radicals formation by the following chain reaction Eq. (1)–(5).
3.2.1. Effect of pHTo study effect of solution pH on the treatment efficiency of the dairy simulated wastewater, the experiments was carried out by adjusting the initial pH of solution from 5 to 9.
Effect of pH on the COD and lactose reduction of the dairy wastewater by O3/UV/H2O2 process was studied at various pH ranges of 5, 7 and 9 as illustrated in Fig. 3 (a) and (b). This result shows that the COD and lactose reduction was low in neutral (pH 7) and alkaline medium (pH 9), and it was protuberant in acidic medium (pH 5). During the treatment process, in higher pH ranges, ozone was continuously oxidized the suspended molecules into dissolved molecules and consequently, ozone demolishes those suspended molecules into small molecules though enhances the COD level in solution [36]. Moreover, decomposition of ozone was quite high at higher pH ranges (7 to 9) subsequently causes a scavenging effect on the solution than pH 5 [37]. In contrast, in acidic medium (pH 5), ozone was directly influenced by the UV radiation and Hydrogen peroxide which was decomposed into OH radicals enhances the COD and lactose reduction. The maximum COD and lactose reduction were attained at a pH value of 5 as 88% and 93.4%, respectively. On comparing the treatment efficiency of the system on dairy wastewater, lactose reduction was very fast than the COD reduction. About 50.0% of lactose reduction and 40.0% of COD reduction was achieved within 30 minutes. Faster and higher lactose removal efficiency and lower and slower COD removal efficiency ensures that fragmentation of lactose takes place initially. But further increase of the COD removal efficiency with time proves the degradation of the fragmentation of bulkier molecules during the reaction.
At different initial pH values, variation in the pH value of dairy wastewater during the treatment with respect to the reaction time were shown in Fig. 3 (c). As we clearly have seen in Fig. 3 (c), although we maintain the initial pH value of wastewater at 5, 7 and 9, within 30 mins of an interval, solution initial pH value was dropped between 3.2 to 2.5. This was due to the formation of organic and inorganic acids during the ozone-based photocatalytic process. In dairy wastewater, lactose was one of the major components which undergoes either degradation or fragmentation during the treatment process. Due to this continuous fragmentation of lactose, it was converted into 4-O-β-galactopyranosyl-D-gluconic acid as shown in Fig. 6 [38]. Although an increase of circulation rate and H2O2 concentration could not much influence in the variation of pH ranges. At the end of the treatment process (180 mins), almost 80% of the lactose was degraded into lactobionic acid. Besides, the increase of treatment time beyond the 3 h not much influence in the variation of pH ranges.
Increasing the circulation rate (50 to 150 mL/h) of the wastewater, solution pH of the wastewater rapidly falls to 2.2 during the treatment process. The same result was observed by maintaining an initial pH of the solution 7 and 9. But increasing the circulation rate of simulated dairy wastewater, the increased time required to fall of solution pH during the course of the reaction.
3.2.2. Effect of circulation rateThe experimental trials were carried out for the simulated dairy wastewater with the circulation rate of 50–150 mL/h at a constant H2O2 of 1mL with different pH levels were shown in Fig. 4 ((a)–(f)).
As can be seen from the Fig. 4 (a) and (b), at pH 5, maximum COD and lactose reduction were attained at a minimum circulation rate of 50 mL/h. Increase of circulation rate from 50 to 150 mL/h clearly showed that the decrease of COD and lactose removal efficiency. Similar results were attained at a pH of 7 and 9 as shown in Fig. 4 ((c), (d), (e) and (f)). On all the pH cases, more than 60% of COD and 70% of lactose reduction was attained at a circulation rate of 50 mL/h and reaction time of 180 mins. From Fig. 4 (a) and (b), At low circulation rate (50 mL/h), the acidic condition (pH 5) improves more than 80% COD and lactose reduction than basic and alkaline conditions of solution within 180 mins. Nevertheless, Fig. 4 ((c), (d), (e) and (f)) reveals that the increase of pH to basic (pH 7) and alkaline (pH 9) condition considerably reduces the COD and lactose reduction about 10 to 15% compared to acidic condition with the same time and circulation rate. The maximum reduction of COD and lactose reduction was may be achieved at the higher circulation rate even at higher pH of 7 and 9 by increasing the reaction time beyond the 180 mins.
It could be clearly explained by the fact that the effluent detention time in the photocatalytic reactor of the dairy wastewater was considerably reduced on increasing the circulation rate from 50 to 150 mL/h. At higher circulation rate, detention time between target molecules and generated oxidative species in the reactor during the treatment process was considerably low which subsequently affects the reduction efficiency of COD and lactose removal. Since the hydroxyl radicals and superoxides were short in life which could not be effectively utilized in the treatment process at the higher circulation rate which could be maximum destruction of oxidative species. Contrariwise, an acquaintance of chemically oxidative species (OH radicals, O3 and HO2) with target molecules of dairy wastewater was maximum at a low circulation rate of 50 mL/h which considerably enhances the COD and lactose reduction efficiency.
3.2.3. Effect of H2O2 dosageDuring the treatment, the H2O2 dosage was varied from 0.5 to 1.5 mL per 100 mL of dairy wastewater with a constant circulation rate of 100 mL/h throughout the process.
Effect of H2O2 dosage on COD and lactose reduction with respect to time was illustrated on the Fig. 5. As from Fig. 5 (a) and (b), the result reveals that the increase of H2O2 dosage from 0.5 to 1.0 mL/h consequently enhances the COD and lactose reduction of 82% and 77% respectively at the pH of 5. Further increase of H2O2 dosage to 1.5 mL decreases the COD and lactose reduction efficiency of 10% and 11% from the H2O2 dosage of 1.0 mL.
Similarly from Fig. 5 ((c), (d), (e) and (f)), the experimental trials were repeated for the dairy effluent with the pH value of 7 and 9 and the result reveals that the decrease of COD (< 88%) and lactose content (< 75%) by way of increasing H2O2 dosage from 1.0 to 1.5 mL/h. From these results, the performance of oxidation process greatly depends upon the formation of the hydroxyl radicals [39].
The supplementary addition of H2O2 into photocatalytic oxidation also enhances the production of more number of hydroxyl radicals. The lowest COD and lactose reduction efficiency at 0.5 mL of H2O2 dosage causes insufficient radical formation during the photocatalytic reaction. Upon increasing the H2O2 dosage from 1.0 mL/h increases the number of reactive radical formation by photochemical synergistic O3/UV/H2O2. Maximum COD and lactose reduction efficiency of 82% and 82.8% at the 1.0 mL/h of H2O2 dosage and pH of 5, confirms the formation of more number of highly reactive radical formation during the course of the reaction. In contrast, a decrease of COD and lactose reduction of 28% and 55.6% at the H2O2 dosage of 1.5 mL and pH of 5. Ineffective utilization of reactive radicals due to the scavenging of hydroxyl radicals during the oxidation process continuously decreases the degradation efficiency of organic matter [40].
This result shows that the lactose and COD reduction ensures that the fragmentation followed by degradation of the organic molecules may take place during the treatment process. Excess amount of H2O2 in wastewater generates a maximum number of hydroxyl radicals which promotes secondary reactants like hydroperoxide instead of increasing the degradation efficiency of the pollutants. This hydroperoxide was less reactive than hydroxyl radicals was unfavor of organic and inorganic removal. It was concluded that the rate of generation of hydroxyl radicals should be controlled by the dosage of H2O2.
Based on the H2O2 dosage on the dairy wastewater, the hydroxyl radicals were formed. At alkaline pH of 9 the ozone was quickly decomposed and rapidly formed more number of hydroxyl radicals than at pH 5 and 7 [39]. Nevertheless, at a higher dosage of H2O2, there was a high risk for H2O2 acted as a radical scavenger which possibly decreases the OH* radicals concentration in the solution as per the following reaction [22] (Eq. (10)–(11))
Lactose was directly related to the COD reduction. During the treatment process, the lactose reacts with hydroxyl radicals and it was partially oxidized into lactobionic acid as a byproduct in COD reduction. The presence of H2O2, OH°, HO2° radicals in dairy effluent during the photocatalytic process, continuously reacts with the carbonyl group of lactose and subsequently degraded into 4-O-β-galactopyranosyl-D-gluconic acid due to the free radical mechanism as shown in Fig. 6 [38]. During this mechanism, presence of secondary oxygen shown in (Eq-5) could be possible to react with lactobionic acid and finally converted into H2O.
This could be further supported by Fig. 7 describes that the electrical conductivity and total dissolved solids (TDS) of the wastewater samples were continuously increased during the treatment process. At the initial stage, the conductivity and TDS of wastewater were measured as 0.5 ms/cm and 230 ppm respectively and it was continuously increased to 2.9 ms/cm and 1,247 ppm respectively. The changes in EC and TDS were explained by the fact that fragmentation of bulkier molecules in the dairy wastewater into smaller ionic species, which may correspondingly, increases the conductivity as well as the TDS.
3.2.4. Optimum conditionsFrom the above sections, the result concluded that the efficiency of COD and lactose reduction was greatly influenced by the initial pH (5 to 9), circulation rate (50–150 mL/h), H2O2 dosage (0.5 to 1.5 mL) and reaction time (30 to 180 mins).
Table S1 presents the comparison results of COD reduction on ozone-based photocatalytic process to other treatment processes. Generally, dairy industry wastewater contains organic mater in which most of the researchers were carried out the biological and physicochemical processes. From table S1, the hybrid AOP process gives more COD reduction in dairy wastewater than other methods. Torres-Sánchez, López-Cervera [41], reveals that the maximum COD reduction of 95% was achieved when combining the AOP process of ozone and fenton into the electrocoagulation process. The present work exposes that the O3/UV/H2O2 system gave better results in a COD reduction of 88% compared to other biological and physicochemical processes. In the future, more than 90% of COD reduction was may be achieved by a combination of the hybrid process of O3/UV/H2O2 and membrane technology.
The synergistic effect of a hybrid process such as O3+H2O2, O3+UV, UV+H2O2 and O3+UV+H2O2 were carried out and its corresponding results were illustrated in Fig. S1 (a) and (b). The hybrid O3+UV+H2O2 process gives the yield of 65% of COD and 52.36% of lactose reduction than other hybrid processes such as O3+H2O2, O3+UV and UV+H2O2 at the pH of 7.
From Fig. S1 (c) and (d), when the treatment time of effluent was increased from 60 to 180 mins, the removal efficiency of COD was increased from 74 to 88% and lactose conversion was raised from 83.6 to 93.4% in the O3+UV+H2O2 process. In all the buffering conditions, the acidic medium (pH 5) was found to be more significant on the COD (88%) and lactose (93.4%) reduction efficiency. Also, the result reveals that the optimum condition for maximum COD and lactose conversion was taking place at an initial pH of 5, circulation rate of 50 mL/h, reaction time of 180 mins and H2O2 dosage of 1 mL.
Based on this optimum condition, the performance of O3/UV/H2O2 process was carried out in simulated and real dairy industrial effluent by measuring the parameters such as COD, BOD, lactose, turbidity and color were presented in Table S2.
The maximum removal efficiency of COD, lactose, turbidity and color were attained by 88–92, 97–98, 97–99% and colorless respectively. During the treatment process, more than 92% of the biological oxygen demand (BOD) was removed in both simulated and real wastewater which represents the elimination of the maximum amount of microorganisms present in dairy effluent.
4. ConclusionIn this research, the synergistic effect of advanced oxidation processes such as ozone, UV radiation; O3/H2O2, O3/UV, UV/H2O2 and O3/UV/H2O2 for treatability of dairy industry wastewater were studied. Effect of different process variables such as initial pH, reaction time, circulation rate and H2O2 dosage was observed. This study exposed that O3/UV/H2O2 system can be a better marginal to other hybrid AOP processes and conventional methods when worked under ideal circumstances.
When conducting experiments with individual processes such as ozone, UV radiation and H2O2, there were no noticeable results (< 10%) that were found in COD and lactose reduction. COD of 32.5%, 35.2%, 25%, 83% and lactose of 43.6%, 38.2%, 80% reduction efficiency were attained during the hybrid AOP process of O3/H2O2, O3/UV, UV/H2O2 and O3/UV/H2O2 processes at a pH of 7 and circulation rate of 50 mL/h, respectively. From these results, the hybrid O3/UV/H2O2 process was attained maximum COD, lactose reduction of 88% and 93.4% respectively under the optimum conditions of pH of 5, reaction time of 180 mins, circulation rate of 50 mL/h and H2O2 dosage of 1.5 mL. Reduction efficiency of COD and lactose decreased by an increase in H2O2 dosage of 1.5 mL, initial pH of 9 and circulation rate of 150 mL/h. This is due to at an alkaline pH and higher dosage of H2O2, the ozone decomposition rate was higher eventually causes a scavenging effect while at maximum circulation rate lack of retention time between radicals and pollutants eventually decreases the COD and lactose reduction efficiency.
However, after the O3/UV/H2O2 treatment process, TDS and EC of treated wastewater were increased to 1,043 ppm and 2.5 ms from its initial level ensures the incomplete/partial degradation of organic pollutants present in the dairy effluent. As of this optimum process/conditions, the real dairy effluent was treated while the final concentrations of COD of 105 mg/lit, BOD of 18 mg/lit, lactose of 13 mg/L were achieved.
Removal efficiency of BOD in real-time dairy effluent satisfies the effluent standards recommended by the Central Pollution Control Board (CPCB) of India. However, TDS and pH of treated effluent did not meet the permissible limits of standards, which need further treatment process before the effluent was to be discharged into receiving bodies.
AcknowledgmentsWe acknowledge that this research work was funded by All India Council for Technical Education (AICTE) – Research Promotion Scheme (RPS), the government of India (order no: 8-208/RIFD/RPS (POLICY-1)/2018-19).
References1. Alimoradi S, Faraj R, Torabian A. Effects of residual aluminum on hybrid membrane bioreactor (Coagulation-MBR) performance, treating dairy wastewater. Chem Eng Process. 2018;133:320–324.
2. Tiwari S, Behera CR, Srinivasan B. Simulation and experimental studies to enhance water reuse and reclamation in India’s largest dairy industry. J Environ Chem Eng. 2016;4:605–616.
3. Jaganmai G, Jinka R. Production of Lipases from Dairy Industry Wastes and its Applications. Int J Curr Microbiol Appl Sci. 2017;5:67–73.
4. Suman A, Ahmad T, Ahmad K. Dairy wastewater treatment using water treatment sludge as coagulant: a novel treatment approach. Environ Dev Sustainability. 2018;20:1615–1625.
5. Falahati F, Baghdadi M, Aminzadeh B. Treatment of dairy wastewater by graphene oxide nanoadsorbent and sludge separation, using In Situ Sludge Magnetic Impregnation (ISSMI). Pollution. 2018;4:29–41.
6. Subramani T, Rajkumar V, Priyanka S. Treatment Of Dairy Waste Water From Salem. In J Appli Innov Eng. 2017;6:263–273.
7. Mejía-Morales C, Hernández-Aldana F, Cortés-Hernández DM, Rivera-Tapia JA, Castañeda-Antonio D, Bonilla N. Assessment of Biological and Persistent Organic Compounds in Hospital Wastewater After Advanced Oxidation Process UV/H2O2/O3. Water Air Soil Pollut. 2020;231:89.
8. Brião VB, Vieira Salla AC, Miorando T, Hemkemeier M, Cadore Favaretto DP. Water recovery from dairy rinse water by reverse osmosis: Giving value to water and milk solids. Resour Conserv Recycl. 2019;140:313–323.
9. Labbé JI, Ramos-Suárez JL, Hernández-Pérez A, Baeza A, Hansen F. Microalgae growth in polluted effluents from the dairy industry for biomass production and phytoremediation. J Environ Chem Eng. 2017;5:635–643.
10. Kolev Slavov A. General characteristics and treatment possibilities of dairy wastewater–A review. Food technol biotech. 2017;55:14–28.
11. Tawfik A, Sobhey M, Badawy M. Treatment of a combined dairy and domestic wastewater in an up-flow anaerobic sludge blanket (UASB) reactor followed by activated sludge (AS system). Desalination. 2008;227:167–177.
12. Martín-Rilo S, Coimbra RN, Martín-Villacorta J, Otero M. Treatment of dairy industry wastewater by oxygen injection: performance and outlay parameters from the full scale implementation. J Clean Prod. 2015;86:15–23.
13. Markou V, Kontogianni MC, Frontistis Z, Tekerlekopoulou AG, Katsaounis A, Vayenas D. Electrochemical treatment of biologically pre-treated dairy wastewater using dimensionally stable anodes. J Environ Manage. 2017;202:217–224.
14. Bassala HD, Kenne Dedzo G, Njine Bememba CB, et al. Investigation of the efficiency of a designed electrocoagulation reactor: Application for dairy effluent treatment. Process Saf Environ Prot. 2017;111:122–127.
15. Tetteh EK, Naidoo DB, Rathilal S. Optimization of photo-catalytic degradation of oil refinery wastewater using Box-Behnken design. Environ Eng Res. 2019;24:711–717.
16. Loures CCA, Samanamud GRL, de Freitas A, Oliveira IS, de Freitas LV, Almeida C. The use of Advanced Oxidation Processes (AOPs) in dairy effluent treatment. American J Theo Appl Statis. 2014;3:42–46.
17. dos Santos Pereira M, Borges AC, Heleno FF, Faroni LRDA, da Silva JCGE. Experimental Design Optimization of Dairy Wastewater Ozonation Treatment. Water Air Soil Pollut. 2018;229:74.
18. Krzemińska D, Neczaj E, Borowski G. Advanced oxidation processes for food industrial wastewater decontamination. J Ecol Eng. 2015;16:61–71.
19. Bruguera-Casamada C, Araujo RM, Brillas E, Sirés I. Advantages of electro-Fenton over electrocoagulation for disinfection of dairy wastewater. Chem Eng J. 2019;376:119975.
20. Bilińska L, Gmurek M, Ledakowicz S. Textile wastewater treatment by AOPs for brine reuse. Process Saf Environ Prot. 2017;109:420–428.
21. Assalin MR, Almeida ES, Rosa MA, Moraes SG, Duran N. Application of ozonation process in industrial wastewaters: textile, kraft E1 and whey effluents. Environ Technol. 2014;25:867–872.
22. Karat I. Advanced Oxidation Processes for removal of COD from pulp and paper mill effluents: A Technical, Economical and Environmental Evaluation [dissertation]. Stockholm: KTH Royal Institute of Technology; 2013.
23. Varga L, Szigeti J. Use of ozone in the dairy industry: A review. Int J Dairy Technol. 2016;69:157–168.
24. Carvalho MCS, Borges AC, Pereira MdS, Heleno FF, Faroni LRD, Campos LC. Combined use of O3/H2O2 and O3/Mn2+ in flotation of dairy wastewater. Revist Ambiente Água. 2018;13.
25. Satoab K, Saito T. A newly developed wastewater treatment by using solidification reaction of milk fats and proteins through ozonation. Chem Eng Trans. 2013;32:1–6.
26. László Z, Kertész S, Mlinkovics E, Hodúr C. Dairy Waste Water Treatment by Combining Ozonation and Nanofiltration. Sep Sci Technol. 2007;42:1627–1637.
27. Zakar M, Kovács I, Muhi P, Lakatos EH, Keszthelyi-Szabó G, László Z. The Effect of Advanced Oxidation Pre-Treatment on the Membrane Filtration Parameters of Dairy Wastewater. Hung J Ind Chem. 2017;45:23–27.
28. Stanisławek E, Kowalik-Klimczak A. Integration of advanced oxidation process with nanofiltration for dairy effluent treatment. Chall Mod Technol. 2017;8:3–6.
29. Melo JMO, Duarte JLS, Ferro AB, Meili L, Zanta CLPS. Comparing Electrochemical and Fenton-Based Processes for Aquaculture Biocide Degradation. Water Air Soil Pollut. 2020;231:79.
30. Leal MCMR, Freire DMG, Cammarota MC, Sant’Anna GL. Effect of enzymatic hydrolysis on anaerobic treatment of dairy wastewater. Process Biochem. 2006;41:1173–1178.
31. Loloei M, Nekonam G, Alidadi H, Kor Y. Study of the coagulation process in wastewater treatment of dairy industries. Int J Environ Health Eng. 2014;3:12.
32. Benazzi TL, Di Luccio M, Dallago RM, Steffens J, Mores R, Do Nascimento MS, Krebs J, Ceni G. Continuous flow electrocoagulation in the treatment of wastewater from dairy industries. Water Sci Technol. 2016;73:1418–1425.
33. APHA. Standard Methods For Examination of Water and Wastewater. WWA; Washington, DC: 2005.
34. Slavov AK. General Characteristics and Treatment Possibilities ofDairy Wastewater - A Review. Food Technol Biotechnol. 2017;55:14–28.
35. Pourgholi M, Jahandizi RM, Miranzadeh M, Beigi OH, Dehghan S. Removal of Dye and COD from textile wastewater using AOP (UV/O3, UV/H2O2, O3/H2O2 and UV/H2O2/O3). J Environ Health Sustain Dev. 2018;621–629.
36. Yasar A, Ahmad N, Chaudhry M, Rehman M, Khan A. Ozone for Color and COD Removal of Raw and Anaerobically Biotreated Combined Industrial Wastewater. Polish J Environ Studies. 2007;16:
37. Manikandan P, Palanisamy PN, Baskar R, Sivakumar P, Sakthisharmila P. Optimization of treatment efficiency of UV/H2O2 process on simulated textile industry wastewater. Desalin Water Treat. 2016;57:27169–27180.
38. Belkacemi K, Hamoudi S. Chemocatalytic Oxidation of Lactose to Lactobionic Acid over Pd–Bi/SBA15: Reaction Kinetics and Modeling. Ind Eng Chem Res. 2010;49:6878–6889.
39. Abreu P, Pereira E, Campos CM, Naves F. Photocatalytic Oxidation Process (UV/H2O2/ZnO) in the treatment and sterilization of dairy wastewater. Acta Sci Technol. 2013;35:75–81.
40. Lucas M, Peres J, Puma G. Treatment of winery wastewater by ozone-based advanced oxidation processes (O3, O3/UV and O3/UV/H2O2) in a pilot-scale bubble column reactor and process economics. Sep Purif Technol. 2010;72:235–241.
41. Torres-Sánchez AL, López-Cervera SJ, de la Rosa C, Maldonado-Vega M, Maldonado-Santoyo M, Peralta-Hernández JM. Electrocoagulation process coupled with advance oxidation techniques to treatment of dairy industry wastewater. Int J Electrochem Sci. 2014;9:6103–6112.
42. Kothari R, Kumar V, Pathak VV, Tyagi VV. Sequential hydrogen and methane production with simultaneous treatment of dairy industry wastewater: Bioenergy profit approach. Int J Hydrog Energy. 2017;42:4870–4879.
43. Bhuyar KD, Suke SG, Dawande S. Treatment of milk wastewater using up-flow anaerobic packed bed reactor. Pol J Chem Technol. 2015;17:84–88.
44. Al-Jabari M, Dwiek H, Zahdeh N, Eqefan N. Reducing Organic Pollution of Wastewater from Milk Processing Industry by Adsorption on Marlstone Particles. Int J Thermal Environ Eng. 2017;15:57–61.
Fig. 3(a) Effect of pH on COD reduction (b) effect of pH on lactose reduction and (c) Variation of initial pH levels with respect to reaction time. 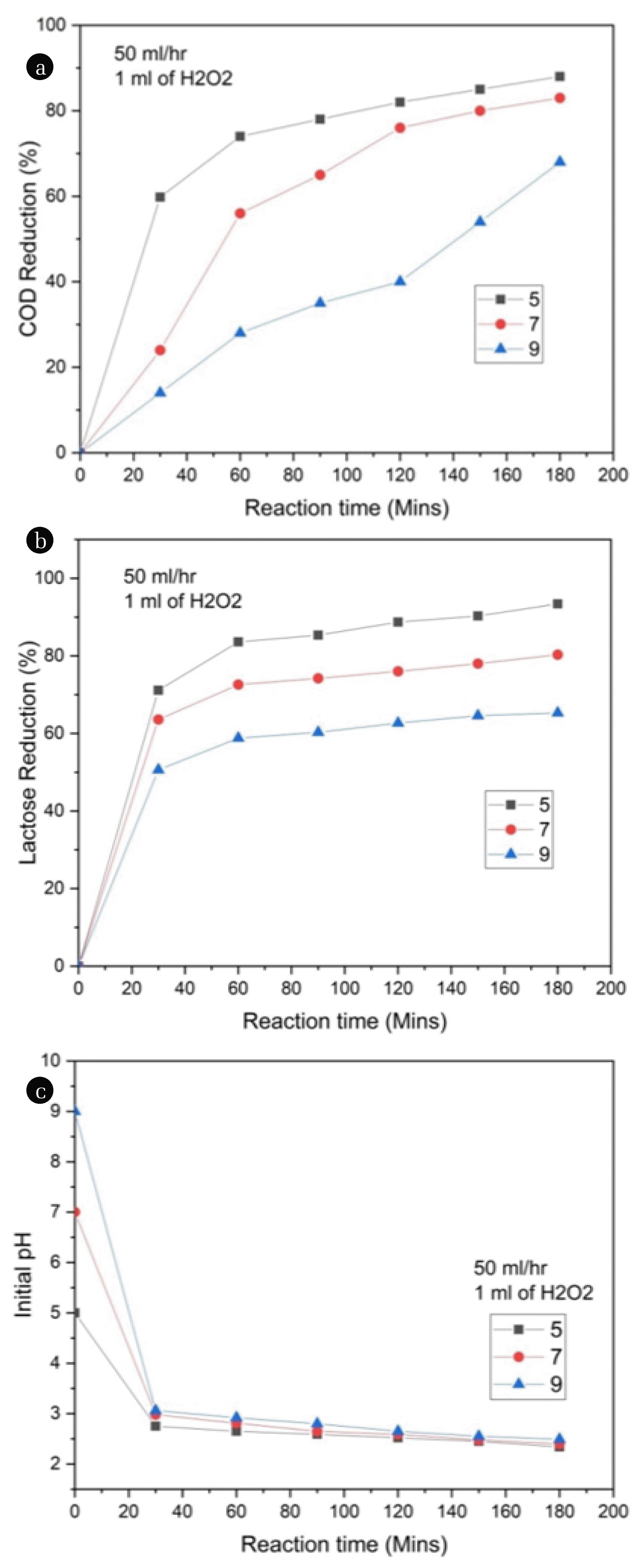
Table 1Characterization of Dairy Wastewater |
|
||||||||||||||||||||||||||||||||||||||||