1. Introduction
Sanitary landfills are used worldwide to dispose municipal solid waste, although it is not the most economical and environmental friendly method [1]. The sanitary landfills should be carefully designed and maintained to minimize the release of contaminants to the environment. While these sites are constructed to dispose waste by burial in land, open dump sites are also use in some countries. Generation of leachate is one of the major problems which occur whether the dump site is open or buried one [2].
Leachate is known as a highly polluted liquid, which comes out from sanitary landfills, when water passing through it. It contains a wide range of chemical compounds such as NO3 −, NO2 −, PO4 3−, NH4 +, heavy metals and oxygen demanding substances. Characteristics of leachate may depend on several factors such as temperature and other climatic factors, degree of wetness, age of landfill and type of waste [3]. Leachate pollutes both water and soil [4] and this would in turn affect the human health. As such, generation of leachate from landfills should control, collect properly and treat before releasing to the environment, wherever possible.
Nitrogen compounds including nitrate (NO3 −), nitrite (NO2 −) and ammonium (NH4 +) can be found in leachate. Total nitrogen (TN) content of leachate can be in the range of 2.6–945 mg/L and the upper limit can be even as high as 1,416 mg/L [5]. Nitrogenous compounds, mainly the compounds which contain inorganic nitrogen, create environmental impacts such as eutrophication, acidification of surface water, toxicity to aquatic animals, and increased algal blooms [6]. Nitrate and nitrite are known as primary water pollutants and regulated in drinking water standards such as the United States Environmental Protection Agency (USEPA) national primary drinking water regulations and World Health Organization (WHO) drinking water quality guidelines. High nitrate levels in drinking water can cause methemoglobanemia in infants and stomach cancer in adults [7].
WHO specifies that the ammonia level occurs in drinking-water is at concentrations well below those of health concern. However, ammonia can cause taste and odor problems when its concentrations are above 35 and 1.5 mg/L, respectively [7]. Amount of the ammonia found in leachate are generally higher than the WHO limits [2]. For example, the ammonia level can be even above 400 mg/L in stabilized landfill leachate [8]. If discharged in to a water body, such high concentrations of ammonia may create pollution issues in receiving environments. Therefore, leachate should be treated before releasing to the environment.
Biological, physical, chemical and physiochemical methods are available for removing nitrogenous compounds from leachate. While consumes low energy, biological treatment methods need long retention times and more space [9]. The physiochemical methods such as ammonia stripping, coagulation, flocculation, precipitation and adsorption can be expensive, mainly due to high cost for chemicals. Membrane technology is also another effective treatment method, but it is expensive and generates concentrated brine solutions [1, 9].
Electrochemical treatment method is a technology which can be used in removing nitrogenous compounds from contaminated water. Electrochemical cell consists of two electrodes which are known as anode and cathode. Anode contributes to the oxidation process and cathode contributes to the reduction process. Electrochemical methods occupy small area and it needs relatively low investment cost. However, to be energy efficient, the electrolyte should have sufficient conductivity. In addition, developing the suitable electrode materials increases the efficiency of the system [10].
Electrochemical removal of nitrogenous compounds can be achieved through electrochemical reduction combined with electrochemical oxidation. Eq. (1)–(3) show the important reactions involved in electrochemical removal of nitrogenous compounds.
Reactions at cathode:
Reactions at anode:
As shown in Eq. (1)–(3), both cathode and anode are important in removing nitrogenous compounds. Although the nitrate and nitrite reduction occurs at cathode, the reduction by-products should be effectively converted to N2 or NO3 − at anode. While N2 leaves the system, NO3 − reduced back to N2 and by-products. These reactions occur in cyclic nature, providing avenues to remove nitrogenous compounds from polluted water. In addition, the presence of chloride ions in electrolyte would enhance performance since electrochemically generated chlorine acts favourably in oxidizing NH4 + [11]. Moreover, through careful selection of electrodes, simultaneous removal of other co-existing pollutants also would be possible (e.g., organics contaminants, micro-organisms) [12].
Electrochemical nitrate removal has been studied and reported in literature [7, 13–17]. Cathode materials such as Ni, Fe, Rh, Pb, and Cu has been researched in literature, and among them, Cu has been identified as one of the best cathodes in electrochemical reduction of nitrate [18–21]. About 99% removal of nitrate from groundwater within 2 h of reaction time has reported in literature using laboratory developed Cu/Ti cathode and IrO2/Ti anode [7]. Conductivity of groundwater in above study was ranged from about 350 μs cm−1 – 1,300 μs cm−1, while the NO3 − concentration was around 175 mg/L. Using electrochemical technology, near complete removal of ammonium from solutions at the presence of chloride ion is reported in literature [22]. More than 95% of ammonia removal from treated municipal wastewater, which were collected after aerobic or anaerobic treatment showed the suitability of technology as a post-treatment nitrate removal step [23].
Although the electrochemical techniques has been investigated in literature for removing nitrogenous compounds including nitrate and ammonium, studies on application of the technology to remove nitrogenous compounds from leachate are limited. Since leachate is a complex pollutant which contains varying amounts of different pollutants, removing nitrogenous compounds of leachate can be a challenging task. In addition, selection of low cost electrodes for cathode and anode is a key factor in actual application, especially in developing world. As such, this study was mainly targeted on removing nitrogenous compounds from leachate using low cost electrodes as cathode and anode. Considering additional benefits at the point of engineering application, simultaneous removal of total organic carbon (TOC) also investigated.
2. Materials and Methods
2.1. Materials
EDTA, formaldehyde, copper nitrate and sulfuric acid were purchased from Sigma-Aldrich. Sodium hydroxide (96%), hydrogen peroxide, sodium sulfate (anhydrous) (99%), and K2SO4 were purchased from Meron Cochin. Nitrite power pillows and nitrate power pillows were used for nitrite and nitrate detection experiments respectively while Nessler regent, mineral stabilizer and polyvinyl alcohol dispersing agent were used to detect ammonia present in leachate which were from HACH. All aqueous solutions were prepared by using deionized water and all chemicals were analytical grade.
2.2. Methods
2.2.1. Electrochemical reactor set-up
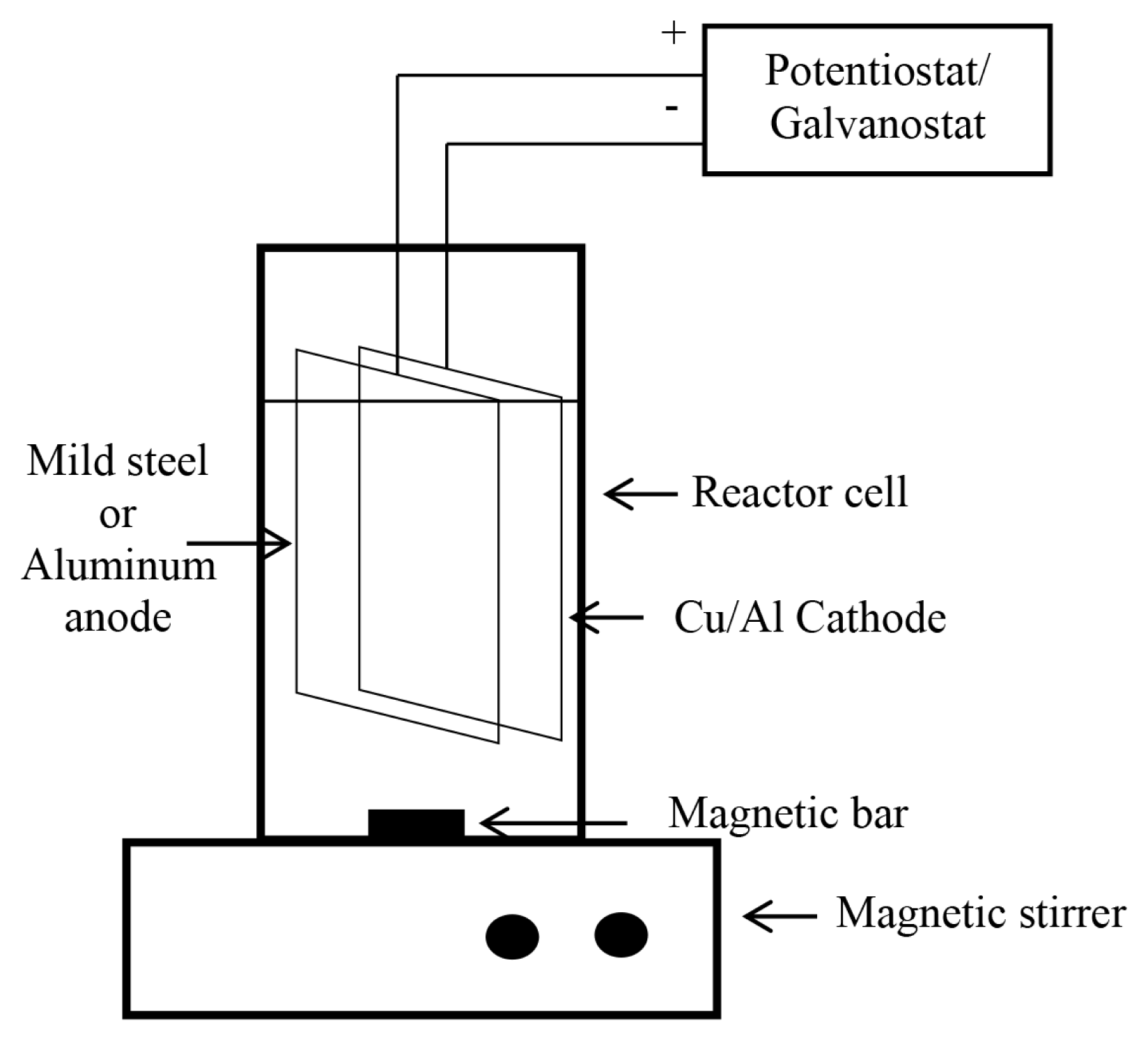
Fig. 1 shows a schematic diagram of the electrochemical cell, which was used to remove nitrogenous compounds and TOC from landfill leachate. A two electrode batch reactor cell with a volume of 50 mL was used as the electrochemical reactor. A potentiostat-galvanostat (Metrohm Autolab PGSTAT 128N) was employed to provide direct current to the reactor. Current was kept at pre-determined required value for necessary reaction duration. Reactions were carried out at room temperature.
The reactor cell shown in Fig. 1 has two electrodes, namely, cathode and the anode. Copper coated Aluminum (Cu/Al) electrodes were prepared at the laboratory using electroless plating and used as the cathode of the electrochemical reactor. In electroless plating, a reducing agent is employed in coating bath to reduce required metal ion on the surface of the substrate material. For coating the cathode, Al sheet was shaped into electrodes of 1 cm × 1 cm. 5% aqueous sodium hydroxide solution at 60°C was used for the removal of grease. Next, the substrate was etched using 5% sulphuric acid and 5% hydrogen peroxide. The operational temperature was maintained at nearly 60°C. Then the substrate was neutralized using 5% sodium hydroxide solution in order to remove etchants that might have remained on the substrate from the previous step. Initial weight of these substrate materials were measured using an electronic scale. The dimensions of the electrodes were measured and their geometrical surface area was calculated. Electroless plating requires a plating bath where the electrode is immersed. In this study EDTA bath was used as the plating bath. EDTA solutions with different concentrations were prepared and sodium hydroxide was added to the solution. The solution was stirred until EDTA completely dissolved. Then 0.05 M cupric nitrate was added to the solution making sure that cupric nitrate is fully dissolved. Solution pH was adjusted as required. Formaldehyde solutions of varied concentrations were used as the reducing agent. Plating was carried out at room temperature, which was around 27°C. The best cathode was selected and subsequently used as the cathode in the electrochemical reactor cell.
Mild steel (MS) or Al (1 cm × 1 cm) was used as anode, considering the low cost of these anode materials.
2.2.2. Electrode characterization
Electrochemical analyses of developed electrodes were carried out using the potentiostat-galvanostat (Metrohm Autolab PGSTAT 128N) equipment. Open circuit potential technique was used in order to analyze the electrode stability. For all these analysis, three electrode systems were used with a Ag/AgCl electrode as the reference electrode (RE). Electrolyte type and other necessary experimental parameters were changed as required and those details are given with relevant figures/illustrations.
Scanning electron microscopy (SEM) was conducted on the coated samples by scanning electron microscope Zeiss Evo LS15. The surface structures of the coated samples were compared with substrate material prior to coating. Surface chemical composition was detected using Energy Dispersive X-ray (EDX) analyzer coupled with the Zeiss Evo LS15 scanning electron microscope.
2.2.3. Leachate collection and characterization
Leachate from Gohagoda (N 70 19′, E 800 37′) and Udapalatha (N 70 09′, E 800 35′) landfill sites were collected and characterized before using the samples for reactor efficiency analysis. Gohagoda landfill is an active landfill while Udapalatha landfill is an abandoned landfill. DR/2010 HACH spectrophotometer was used for detecting NH3, NO3 − and NO2 − in leachate before and after treatment. Total Carbon Analyzer (Shemadzu TOC-L CSH 638-91111-48) was used to find the total carbon (TC), total inorganic carbon (IC), TOC and TN amount present in the leachate samples before and after treatments. pH and electrical conductivity were measured using appropriate probes.
3. Results and Discussion
3.1. Cathode Material Development
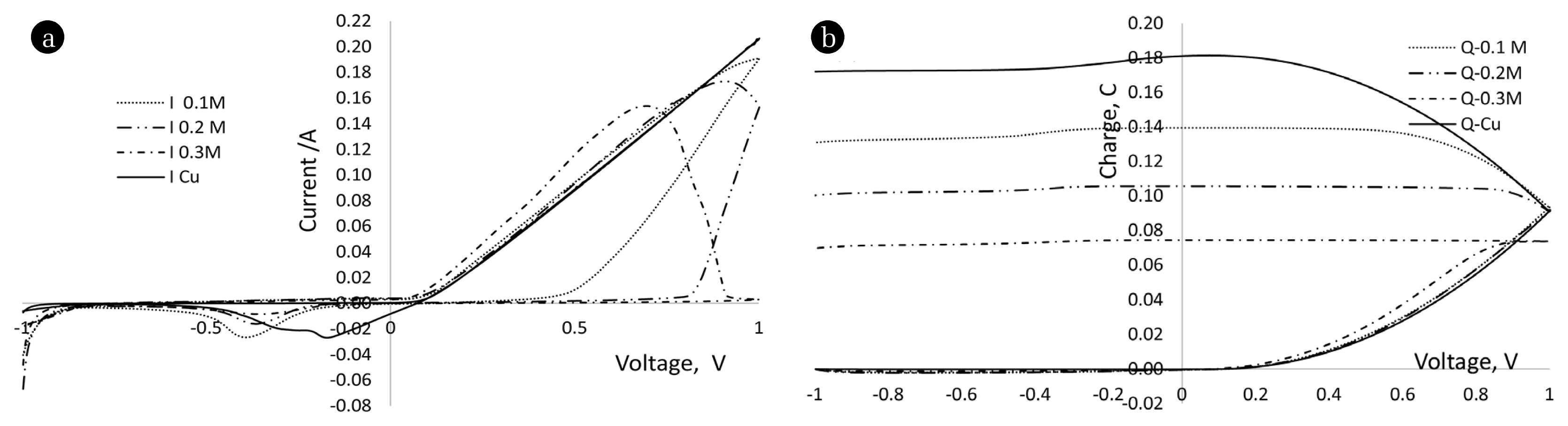
Electroless plating was carried out to coat Cu on Al substrates at varying coating conditions. The coating layers were analyzed using SEM and EDX technologies. SEM images of the electrodes and results of the EDX analysis are shown in Fig. 2 and Table 1, respectively. Fig. 2 and Table 1 clearly reveal that the electroless coating technique has successfully introduced Cu on to the Al plate electrodes. Table 1 shows that the coated Cu percentage is almost the same irrespective of the concentration of the reducing agent, i.e., HCOH. In addition, Fig. 2 shows that the surfaces of developed Cu/Al cathodes may contain higher amount of pores compared to the uncoated Cu and Al cathodes. The laboratory developed cathode surfaces contain cracks and grains developed on them as a result of Cu deposition. These structural changes would be important to get higher electrochemically active surface area on electrodes. The electrochemically active surface areas of the electrodes can be indirectly compared through cyclic voltametric (CV) analysis. Therefore, CV analysis was carried out using Cu/Al electrodes and Cu plate as working electrode in a three electrode cell setup. Fig. 3 shows the obtained current vs. potential and charge vs. potential curves. It can be seen that the uncoated Cu electrode shows the highest charge compared to the other three electrodes. Since charge is an indirect measure of electrochemical active surface area [7], it can be said that the uncoated electrode contains a higher electrochemically active surface area compared to other three electrodes. Although this finding is contradictory to the simple visual observation of SEM images, electrochemical analysis would be more accurate than visual observation. Thus, it is believed that the electrochemically active surface area is higher in uncoated Cu plate compared to the three Cu/Al coated electrodes.
Since cathode plays a crucial role in denitrification, four electrodes were used in removing nitrate from a synthetic nitrate solution. The reason behind conducting this investigation is only to compare the performance of the four cathodes under consideration. Titanium anode was used in this investigation in order to minimize the involvement of anode in electrocoagulation; it should be noted that both Al and MS anodes would contribute in removing nitrate, due to their ability in developing coagulants during reaction. Results of this analysis are reported in Table 2.
Table 2 shows that, although the electrochemically active surface area is large, the removal efficiency of pure Cu cathode is lower than that of the developed electrodes. This finding can be explained as follows. As shown in SEM images (Fig. 2), the developed electrodes contain defects on the surface. As such, during large reaction durations, migration of electrolyte solution in to the deposited Cu layer may occur. The migrated electrolyte would form minute electrolysis cells within the electrode coating layer. Thus, there is a possibility of formation of anodic sites inside the deposited Cu layers [7]. Since the Cu layers are deposited on Al substrates, electrocoagulation process may occur during the reaction due to the anodic reaction at Al surface. The anodically generated Al3+ ions support the electrocoagulation [24, 25]. In literature, it has been reported that electrocoagulation process does support the nitrate removal [26].
Based on above results, Cu/Al cathode coated with 0.1 M HCOH was selected for further investigations. Subsequently, few more experiments were carried out to identify the coating bath pH vs. coating morphology/quality. Fig. 4 shows the surface morphology of the cathodes developed at different pH (all the other plating bath conditions were kept the same). Based on the surface morphology (visual observation alone), it can be said that the coating developed at pH 13 is better than the other coating layers.
3.2. Removing Nitrogenous Compounds from Leachate
3.2.1. Efficiency studies with actual leachate samples
Characteristics of leachate from two different landfills in Sri Lanka are shown in Table 3. Initially, landfill leachate sample which was collected from Gohagoda landfill site was treated in electrochemical reactor with Cu/Al cathode. Either Al or MS was used as the anode of the reactor. TN removal as well as TOC removal was studied, since the simultaneous removal of TOC would be an added advantage. Results of this investigation are shown in Table 4.
As shown in Table 4, electrochemical reactor has the possibility of simultaneously removing TN as well as TOC. This study is beneficial since it provides additional details about removing major co-existing pollutants. It can be seen that the Al anode together with Cu/Al cathode gives better performance in removing TN. However, when it comes to removing TOC, MS anode gives better performance.
Al anode is good in electrocoagulation process and it has been reported that the electrocoagulation supports denitrification through removing nitrate from the polluted water [26]. Open circuit potential (OCP) study shows OCP of Al and MS electrodes as −1.175 V and −0.96 V, respectively. That means, Al anode shows lower stability compared to MS anode in electrochemical cell. Relatively low stability of Al anode, as evident in OCP test, creates an environment which supports corrosion of the anode and indicates the ability of easily releasing Al3+ in to the bulk solution, compared to releasing Fe3+ ions from MS anode. Therefore, the Al anode shows better performance in electrocoagulation compared to MS anode (It should be noted that Fe based anodes do contribute to the electrocoagulation process). It is reported in literature that electrocoagulation with aluminum anode could achieve better ammonia nitrogen removal compared to iron anode and when the chloride ions exist in the electrolyte, the electrocoagulation exhibits better ammonia nitrogen removal through the generation of active chlorine [27].
However, the same fact would reduce the current efficiency in oxidizing TOC at Al anode, and therefore, the TOC removal efficiency of the Al anode is lower compared to the MS anode. The TOC removal is occurred mainly through electrochemical oxidation. The oxidants generated at the Al anode may have heavily used in corrosion (since the electrode is highly prone to corrosion as shown in OCP results) rather than contributing to oxidation [12]. Moreover, the electro-Fenton process in the cell with Fe base anode would have contributed to the better TOC removal at MS anode [28, 29].
About 80% reduction in TN from synthetic wastewater solution using electrochemical reactor with Fe cathode and a Ti/IrO2-Pt anode in 3 h reaction time is reported [15]. Current density as of the study is reported as 20 mA/cm2; however, the initial TN concentration is around 100 mg/L, which is lower than that of the current study. Compared to the denitrification efficiencies obtained using groundwater, which is 99% [7], the values reported in Table 4 for TN removal are small. This can be due to few reasons. The electrolyte in this study is a complex one with several co-existing pollutants such as organic matter. Applied current is used for redox reactions, however, the efficiency is not fully utilized by the intended reaction. Part of the applied energy is utilized for reactions such as organic carbon removal. TN concentration of this study is far beyond (around 600 mg/L) the other studies stated above (varies between 15–175 mg/L). Therefore, direct comparison of efficiencies is not fair.
Therefore, it can be concluded that, use of Al anode with developed Cu/Al cathode is the best possible combination of electrodes for treating leachate, considering removal of TN from leachate. The same anode-cathode system facilitates simultaneous removal of TOC from leachate as well. In addition, it should be noted that the selected electrodes are of low cost, reducing the initial and maintenance cost of the system.
After selecting the electrodes, further investigations were carried out using Udapalatha landfill leachate. Based on Table 3, pH of both landfill sites were approximately the same. Amount of ammonia-N and TN of Udapalatha leachate was about 50% lower than that of Gohagoda leachate. However, nitrate-N amount of Udapalatha leachate was about 30% higher than that of Gohagoda leachate. TOC amount of Gohagoda leachate was about 30% higher than that of Udapalatha leachate while Nitrite-N amount of both landfill sites were under detectable level.
Udapalatha raw leachate (i.e. as received) was treated under the same experimental conditions mentioned at Table 4. TN and TOC removal efficiencies were found as 31.27% and 26.05%, respectively. Irrespective of the differences mentioned about the two leachate samples, TN and TOC removal efficiencies of two landfill sites were very similar to each other. Further, ammonia-N and nitrate-N removal efficiencies were found out as 22.71% and 91.8%, respectively. These results suggest that the electrochemical reactor is very efficient in denitrification. This may be due to the high activity of the cathode of the reactor in reducing nitrate and support of anode to remove nitrate through electrocoagulation. However, reactor performance in removing ammonia-N is not very high and thus the TN removal efficiency also not very appealing. The electrochemical removal of ammonia occurs due to the electrochemical oxidation which occurs at anode. The low activity of anode towards electrochemical oxidation would have contributed to the low ammonia removal efficiency of the reactor. Nitrite-N amount was very low and couldn’t be able to detect using the spectrophotometer.
The TN removal efficiencies (which are around 30%) reported above suggests that the electrocoagulation may not be able to remove nitrogen to a greater extent, without compromising the energy efficiency of the system. That is, if further removal is essential, the reactions would have to be carried out for extended durations. However, such alternatives will increase the energy requirements making the technology unacceptable. Therefore, this technology would be suitable as a pre-treatment or post-treatment step in a treatment plant for treating surface leachate.
3.2.2. Efficiency studies with diluted leachate samples
In order to further analyze the treatment efficiency at low concentrations, Udapalatha leachate was diluted into 1:4 proportion (leachate: total volume) and investigations were continued. As a result of dilution, EC of the sample was dropped to 2.169 mS.
Table 5 shows the results obtained using diluted leachate. As shown in Table 5, higher current densities do not have supported the removal efficiency. While optimization experiments are required to find the optimum current density, high current densities do not support removing nitrogenous compounds may be due to loss of activity of anode due to high floc formation. As shown in Table 5, at high current density, anode was covered by the flocs. This may be due to the high rate of Al3+ production at anode and lower mass transport rate. This passivation can reduce the activity of electrode and, as a result, reduce the efficiency.
With diluted leachate, around 88% removal of TN was achieved with a current density of 20 mA/cm2 and 6 h reaction duration. Extending the reaction duration did not show any further improvement in reactor efficiency. After achieving high efficiencies in reactor, lower residual amount of pollutants may undergo mass transfer limitations and thus further improvement in efficiency may be limited. Final nitrate levels were under the detection limit of the equipment, suggesting near complete removal of nitrates from diluted leachate. As shown in Table 5, about 30% of TOC removal is achieved in the system. Dilution did not enhance the TOC removal efficiency of the system, suggesting low activity of Al anode in oxidation of organics and possible mass transfer limitations after dilution. The above findings suggest that the present electrochemical reactor is capable of removing nitrogenous compounds to a significantly low level from leachate, if the leachate is pre-treated.
4. Conclusions
Application of electrochemical technologies in removing nitrogenous compounds from landfill leachate was investigated in this research. Cathode material of the reactor was successfully developed using electroless plating technique. Laboratory developed Cu/Al cathode showed good performance in denitrification compared to commercially available Cu plate electrode. Although the electrochemically active surface area of Cu/Al electrode is lower compared to that of Cu plate electrode, contribution from electrocoagulation due to the presence of Al would have increased the efficiency.
Al and MS anodes were incorporated to the electrochemical cell in order to investigate the efficiency of the electrochemical reactor in removing nitrogenous compounds. Developed Cu/Al electrode was used as the cathode material. Al anodes showed promising results compared to MS. Low OCP of Al compared to MS suggested that the Al is more prone to release Al3+ ions to the electrolyte and thus efficient in electrocoagulation compared to MS. It is concluded that the comparatively high efficiency of Al in electrocoagulation positively contributed in removing nitrate from the system and thereby increase the removal efficiency in nitrogenous compounds from leachate. In addition to removing nitrogenous compounds, Al showed moderate performances in simultaneously removing TOC from leachate, providing additional advantage.
When the leachate as received was used to test the reactor performances, high removal of nitrate-N (about 90%) was achieved. However, the removal of ammonia-N and therefore the removal of TN were low (around 30%). It is concluded that this phenomena can be due to the low oxidation ability of Al anode. However, with diluted leachate, a TN removal efficiency of about 88% was achieved. Therefore, the developed electrochemical reactor system can be used as a secondary or tertiary treatment step in a surface leachate treatment plant.